Volume 53 - Issue 2 - April 2005
Research Article
Surface enthalpy of goethite
- Lena Mazeina, Alexandra Navrotsky
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 113-122
-
- Article
- Export citation
-
High-temperature oxide-melt solution calorimetry and acid-solution calorimetry were used to determine the heat of dissolution of synthetic goethite with particle sizes in the range 2–75 nm and measured surface areas of 30–273 m2/g (27–240 × 103 m2/mol). Sample characterization was performed using X-ray diffraction, Fourier transform infrared spectroscopy, the Brunauer, Emmett and Teller method and thermogravimetric analysis. Water content (structural plus excess water) was determined from weight loss after firing at 1100°C. Calorimetric data were corrected for excess water assuming this loosely adsorbed water has the same energetics as bulk liquid water. The enthalpy of formation was calculated from calorimetric data using enthalpies of formation of hematite and liquid water as reference phases for high-temperature oxide-melt calorimetry and using enthalpy of formation of lepidocrocite for acid-solution calorimetry. The enthalpy of formation of goethite can vary by 15–20 kJ/mol as a function of surface area. The plot of calorimetric data vs. surface area gives a surface enthalpy of 0.60±0.10 J/m2 and enthalpy of formation of goethite (with nominal composition FeOOH and surface area = 0) of −561.5±1.5 kJ/mol. This surface enthalpy of goethite, which is lower than values reported previously, clarifies previous inconsistencies between goethite-hematite equilibrium thermodynamics and observations in natural systems.
Adsorption of volatile organic compounds onto porous clay heterostructures based on spent organobentonites
- Lizhong Zhu, Senlin Tian, Yao Shi
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 123-136
-
- Article
- Export citation
-
Model spent cetyltrimethylammonium bromide (CTMAB)-bentonite, and cetyl pyridinium chloride (CPC)-bentonite used for sorbing p-nitrophenol (PNP) from wastewater, as well as virgin CTMAB-bentonite and CPC-bentonite, were employed as the starting materials to prepare porous clay heterostructures (PCHs). The BET surface areas and total pore volumes of the PCHs based on these spent and virgin organobentonites (PNP-CTMAB-PCH, CTMAB-PCH, PNP-CPC-PCH and CPC-PCH) are 661.5 m2/g and 0.25 cm3/g, 690.4 m2/g and 0.27 cm3/g, 506.3 m2/g and 0.30 cm3/g, and 525.4 m2/g and 0.30 cm3/g, respectively. These values approximate those of activated carbon (AC), at 731.4 m2/g and 0.23 cm3/g, and are much larger than those of bentonite and CTMAB-bentonite, at 60.9 m2/g and 0.12 cm3/g, and 3.7 m2/g and 0.0055 m2/g, respectively. The PCHs have slightly higher adsorption capacities for benzene and CC14 than AC at higher relative pressures despite their comparatively lower benzene and CC14 adsorption capacity at lower relative pressures. The existence of PNP in organobentonites also enhances the volatile organic compounds (VOCs) adsorption capacity of PCHs at lower adsorbate concentrations, although some adsorption capacity is lost at higher concentrations. The hydrophobicity order of the adsorbents is: CTMAB-bentonite > AC > PCHs > bentonite. The micro- to mesoporous pore sizes, superior VOC adsorption properties, thermal stability to 750°C and hydrophobicity and negligible influences of PNP on PCHs make spent PNP-containing organobentonites ideal starting materials for synthesis of PCHs and especially attractive adsorbents for VOC sorption control.
Sorption of 3-amino-1,2,4-triazole and Zn(II) onto montmorillonite
- Jaslin Ikhsan, John D. Wells, Bruce B. Johnson, Michael J. Angove
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 137-146
-
- Article
- Export citation
-
Acid-base titrations and attenuated total reflectance-infrared (ATR-IR) spectroscopy of solutions containing Zn(NO3)2 and the herbicide 3-amino-1,2,4-triazole suggested that soluble complexes ZnL2+ and Zn(OH)L+ form, where L represents aminotriazole. Sorption experiments and modeling in systems containing K-saturated Wyoming (SWy-K) montmorillonite suggest that at low concentrations the aminotriazole sorbs primarily in cationic form via an ion-exchange mechanism. Sorption isotherms for aminotriazole are ‘s’-shaped, indicating a co-operative sorption mechanism as the concentration of the molecule increases. At higher concentrations, ATR-IR spectroscopy indicated the presence of cationic and neutral triazole molecules on the surface, while X-ray diffraction data suggest interaction with interlayer regions of the clay. When the concentration of the herbicide was high, initial sorption of aminotriazole cations modified the clay to make the partitioning of neutral molecules to the surface more favorable. Experiments conducted in the presence of Zn(II) indicated that below pH 7, Zn(II) and aminotriazole compete for sorption sites, while above pH 7 the presence of Zn(II) enhances the uptake of aminotriazole. The enhancement was attributed to the formation of an inner-sphere ternary surface complex at hydroxyl sites (SOH) on crystal edges, having the form [(SOZn(OH)L)]0.
Atomic force microscopy study of montmorillonite dissolution under highly alkaline conditions
- Shingo Yokoyama, Masato Kuroda, Tsutomu Sato
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 147-154
-
- Article
- Export citation
-
Montmorillonite dissolution under highly alkaline conditions (pH = 13.3; I = 0.3 M) was investigated by bulk dissolution methods and in situ atomic force microscopy (AFM). In bulk dissolution experiments, initial SiO2 concentrations were high, and a steady state was reached after 136 h. The dissolution rates derived from the edge surface area (ESA) at the steady-state condition at 30, 50 and 70°C were 3.39 x 10−12, 1.75 × 10−11 and 5.81 × 10−11 mol/m2 s, respectively. The AFM observations were conducted under three conditions: (Run I) short-term in situ batch dissolution at RT; (Run II) long-term in situ flow-through dissolution at RT; and (Run III) long-term batch dissolution at 50°C. The observed reductions in montmorillonite particle volume for Runs I and II were due primarily to edge-surface dissolution. The ESA-based dissolution rate for Run I (10−9 mol/m2 s) was three orders of magnitude faster than that for Run II (10−12 mol/m2 s). The rate obtained for Run II corresponded to the rate at the steady-state conditions in the bulk dissolution experiments. A small number of etch pits developed in Run III slightly increased the ESA of montmorillonite since most of the montmorillonite particles were separated into monolayers lacking three-dimensional periodicity. The ESA-based dissolution rate for Run III was 2.26 × 10−11 mol/m2 s. Dissolution rates based on long-term AFM observations could be directly compared with steady-state rates obtained from bulk dissolution experiments. The AFM observations indicated that dissolution occurred at edge surfaces; therefore, the ESA should be used to calculate the dissolution rate for montmorillonite under alkaline conditions. Dissolution rates of individual particles with different morphologies estimated by AFM were similar to rates estimated from bulk dissolution experiments.
Conversion of chrysotile to a magnesian smectite
- Michael Cheshire, Necip Gũven
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 155-161
-
- Article
- Export citation
-
Chrysotile from Thetford Mines in Quebec, Canada was treated first with mild formic or oxalic acid at concentrations of 0.5 to 2.0 N at 200°C in Teflon-lined 12.0 mL Parr bombs. The reaction products were identified by X-ray diffraction as a poorly crystalline Fe-bearing kerolite-like 2:1 layer silicate (which will be described as a kerolitic precipitate or a kerolitic mesophase in this report). Electron microscopic examination showed a thin foily morphology for this kerolitic mesophase that may have formed by the following reaction:(1)
$\mathop {{\rm{M}}{{\rm{g}}_{\rm{6}}}{\rm{S}}{{\rm{i}}_{\rm{4}}}{{\rm{O}}_{{\rm{10}}}}{{\left( {{\rm{OH}}} \right)}_{{8_{\left( {\rm{s}} \right)}}}}}\limits_{{\rm{chrysotile}}} + 6.02{{\rm{H}}^{\rm{ + }}}_{\left( {{\rm{aq}}} \right)} + 0.54{\rm{F}}{{\rm{e}}^{{\rm{2 + }}}}_{\left( {{\rm{aq}}} \right)} \to \mathop {\left( {{\rm{M}}{{\rm{g}}_{{\rm{2}}{\rm{.46}}}}{\rm{Fe}}_{{\rm{0}}{\rm{.54}}}^{{\rm{2 + }}}} \right){\rm{S}}{{\rm{i}}_{\rm{4}}}{{\rm{O}}_{{\rm{10}}}}{{\left( {{\rm{OH}}} \right)}_{\rm{2}}} \cdot n}\limits_{{\rm{kerolitic}}\;{\rm{mesophase}}} {{\rm{H}}_{\rm{2}}}{{\rm{O}}_{\left( {\rm{s}} \right)}} + {\rm{3}}{\rm{.55M}}{{\rm{g}}^{{\rm{2 + }}}}_{\left( {{\rm{aq}}} \right)}$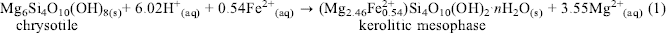
The magnetite impurity in the initial chrysotile asbestos served as the source of Fe in the above reactions. Subsequently, this kerolitic precipitate was reacted with 0.2 N NaOH for 48–96 h at 200°C and a highly crystalline smectite was formed with the same foily morphology as the kerolitic precipitate. X-ray spectral analyses of the kerolitic mesophase and smectite suggest the following reaction to have taken place:(2)
$\eqalign{ & \mathop {\left( {{\rm{M}}{{\rm{g}}_{{\rm{2}}{\rm{.46}}}}{\rm{Fe}}_{{\rm{0}}{\rm{.54}}}^{{\rm{2 + }}}} \right){\rm{S}}{{\rm{i}}_{\rm{4}}}{{\rm{O}}_{{\rm{10}}}}{{\left( {{\rm{OH}}} \right)}_{\rm{2}}}}\limits_{{\rm{kerolitic}}\;{\rm{mesophase}}} \cdot n{{\rm{H}}_{\rm{2}}}{{\rm{O}}_{\left( {\rm{s}} \right)}} + {\rm{0}}{\rm{.53M}}{{\rm{g}}^{{\rm{2 + }}}}_{\left( {{\rm{aq}}} \right)} + 0.54{\rm{NaO}}{{\rm{H}}_{\left( {{\rm{aq}}} \right)}} + 0.01{\rm{F}}{{\rm{e}}^{{\rm{3 + }}}}_{\left( {{\rm{aq}}} \right)} \to \cr & \;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\;\mathop {\;\;{\rm{N}}{{\rm{a}}_{0.54}}\left( {{\rm{M}}{{\rm{g}}_{{\rm{2}}{\rm{.99}}}}{\rm{Fe}}_{{\rm{0}}{\rm{.01}}}^{{\rm{2 + }}}} \right)\left( {{\rm{S}}{{\rm{i}}_{{\rm{3}}{\rm{.46}}}}{\rm{Fe}}_{{\rm{0}}{\rm{.54}}}^{{\rm{3 + }}}} \right){{\rm{O}}_{10}}{{\left( {{\rm{OH}}} \right)}_{{\rm{2}}\;\left( {\rm{s}} \right)}}}\limits_{{\rm{saponite}}} + 0.54{\rm{S}}{{\rm{i}}^{{\rm{4 + }}}}_{\left( {{\rm{aq}}} \right)} \cr} $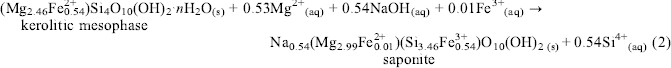
The reaction products, a kerolitic mesophase and smectite, possess a non-fibrous habit in contrast to the fibrous (asbestiform) morphology of chrysotile.
Characteristics of the minerals associated with gold in the Shewushan supergene gold deposit, China
- Hanlie Hong, Liyun Tie
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 162-170
-
- Article
- Export citation
-
The mineralogical properties of goethite and clay minerals from the Shewushan supergene gold deposit have been studied using X-ray diffraction (XRD) and transmission electron microscopy (TEM). These results show that in the weathering zone of the Shewushan supergene gold deposit the mineral assemblage is mainly composed of quartz, kaolinite, halloysite, minor illite and goethite. The coexistence of these minerals is apparently indicative of weak laterization. The Al content in goethite from XRD data is ∼10.0%, suggesting formation by weak desilicification. Observation by TEM shows that the flakes of clay minerals with larger particle size usually have extremely rounded outlines, indicating intensive dissolution of clay minerals. Halloysite is derived from the decomposition of kaolinite and the micrographs of curling, tubular, and club-shaped halloysite strongly suggest significant hydration and thus a water-saturated environment. Both the XRD data of goethite and the micrographs of the clay minerals show that the environment in Shewushan is characteristic of high [H2O] activity and high [SiO2] activity. The high dynamic hydraulic conditions may facilitate the downward migration of the primary gold particles during their mechanical concentration, resulting in the accumulation of gold in the lower portion near the water table.
Modeling the response of pyrophyllite interlayer to applied stress using steered molecular dynamics
- Dinesh R. Katti, Steven R. Schmidt, Pijush Ghosh, Kalpana S. Katti
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 171-178
-
- Article
- Export citation
-
Pyrophyllite is the precursor to other smectite-group minerals which exhibit swelling. The mineral structure of pyrophyllite can lead to other minerals in the smectite group, including montmorillonite, through appropriate isomorphous substitutions. In this work, an atomic model of the pyrophyllite interlayer was constructed. The response of the interlayer was evaluated using steered molecular dynamics simulations. In steered molecular dynamics, external forces were applied to individual atoms to study the response of the model to applied forces. In this work, forces are applied to the surface clay atoms to evaluate the displacement vs. applied stress in the interlayer between clay layers. This paper describes the construction of the model, the simulation procedure, and the results of the simulations which show that under the applied loading, deformation occurs mainly in the interlayer. The clay layers show relatively little deformation. The results show that the relationship between applied stress and displacement of the interlayer is linear. The stress-deformation relationship for the interlayer is presented.
Cation-site partitioning in Ti-rich micas from Black Hill (Australia): A multi-technical approach
- Emanuela Schingaro, Fernando Scordari, Ernesto Mesto, Maria Franca Brigatti, Giuseppe Pedrazzi
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 179-189
-
- Article
- Export citation
-
The crystal chemistry of Ti-rich trioctahedral micas of plutonic origin, cropping out at Black Hill (South Australia) has been investigated by combining electron microprobe analysis, single crystal X-ray diffraction, Mössbauer spectroscopy and X-ray photoelectron spectroscopy. Chemical analyses have shown the samples taken to be quite homogeneous and Ti-rich (TiO2 ≈ 7 wt.%). Mössbauer investigation yielded Fe2+/Fe3+ ≈ 30. X-ray photoelectron spectroscopy analysis seems to suggest the occurrence of three Ti species: octahedral Tr4+(60%), octahedral Ti3+(26%), and tetrahedral Ti4+(14%). The analyzed sample belongs to the 1M polytype and the relevant crystal data from structure analysis are: a = 5.347(1) Å, b = 9.261(2) Å, c = 10.195(2) Å, β = 100.29°(1). Anisotropic structure refinement was performed in space group C2/m, and converged at R = 2.62, Rw = 2.80. Structural details (the c cell parameter, the off-center shift of the M2 cation towards 04, the bond-length distortions of the cis-M2 octahedron, the interlayer sheet thickness, the projection of K−O4 distance along c*, the difference <K−O>outer-<K−O>inner) support the occurrence of the Ti-oxy substitution (VIR2+ + 2(OH)− ⇌ Ti4+ + 202− + H2) in the sample. Analysis of structural distortions as a function of the Ti content revealed that the positions of the oxygens 03 and 04 are displaced in opposite senses along [100]. This produces an enlargement of the M1 site with respect to the M2 site and a shortening of the interlayer distance. This trend seems to be in common with other Ti-rich 1M micas of plutonic origin.
The 2M1 dioctahedral mica polytype: A crystal chemical study
- Maria Franca Brigatti, Daniele Malferrari, Marco Poppi, Luciano Poppi
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 190-197
-
- Article
- Export citation
-
The structure of dioctahedral true micas such as muscovite and celadonitic muscovite (2M1 polytype, space group C2/c) is mostly affected by variations of the octahedral Al (VIAl) content. Crystals with greater Mg, Fe substitutions (i.e. celadonitic muscovite) reduce the dimensional difference between the larger trans-oriented M1 site and smaller cis-oriented M2 octahedral site. The octahedral anionic position O4 is displaced from the center of the hexagon, defined by 031 and 032 oxygen atoms (i.e. ‘octahedral hexagon’), both on and off the (001) plane. The distance between interlayer cation A and O4 is smaller in more substituted species, thus providing different orientations of the O4−H vector, as a function of VIAl. Octahedral distances (<M2−O3> and <M2−O4> are expressed as a function of cell parameters and VIA1 content, thus allowing an approximate estimate of site dimensions. These approximations are useful when a detailed structural refinement is not available. In celadonitic muscovite, the octahedral hexagon mean edge (<O31−O32>Hex) is not significantly affected by VIA1 content. The VIA1 increase produces both a decrease in cell lateral dimensions and a distorted ‘octahedral hexagon’. The decrease in a and b is consistent with a decrease of <O31−O32>Hex, whereas the distortion of the’ octahedral hexagon’ is consistent with an increase of (<031–032>Hex), because an irregular hexagon produces a longer mean edge than a regular hexagon of equal area.
The tetrahedral mean basal edge (VI<O−O>bassal) is reduced as celadonitic substitution progresses. The tetrahedral rotation angle, α was thus found to increase from celadonite to muscovite. However, in muscovite with VIAl content between 1.8 and 2.0 atoms per formula unit (a.p.f.u.), α approaches a saturation value, thus showing a proportional increase of tetrahedral and octahedral sheet lateral dimensions. Furthermore, α variation allows a coarse approximation of the threshold VIAl content, below which celadonitic substitution may not progress.
Announcement
Forthcoming papers
-
- Published online by Cambridge University Press:
- 01 January 2024, p. 198
-
- Article
-
- You have access Access
- Export citation