4 results
Surveillance of healthcare-onset clinical cultures using whole-genome sequencing reveals hidden nosocomial transmission
- Mohamad Sater, Emma Briars, Connor Parrish, Ian Herriott, Kathleen Quan, Keith Madey, Julie Shimabukuro, Linda Dickey, Shruti Gohil, Alfred Wong, Talia Hollowell, Allison Brookhart, Alison Gassett, Miriam Huntley, Susan Huang, Cassiana E. Bittencourt
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 3 / Issue S2 / June 2023
- Published online by Cambridge University Press:
- 29 September 2023, pp. s83-s84
-
- Article
-
- You have access Access
- Open access
- Export citation
-
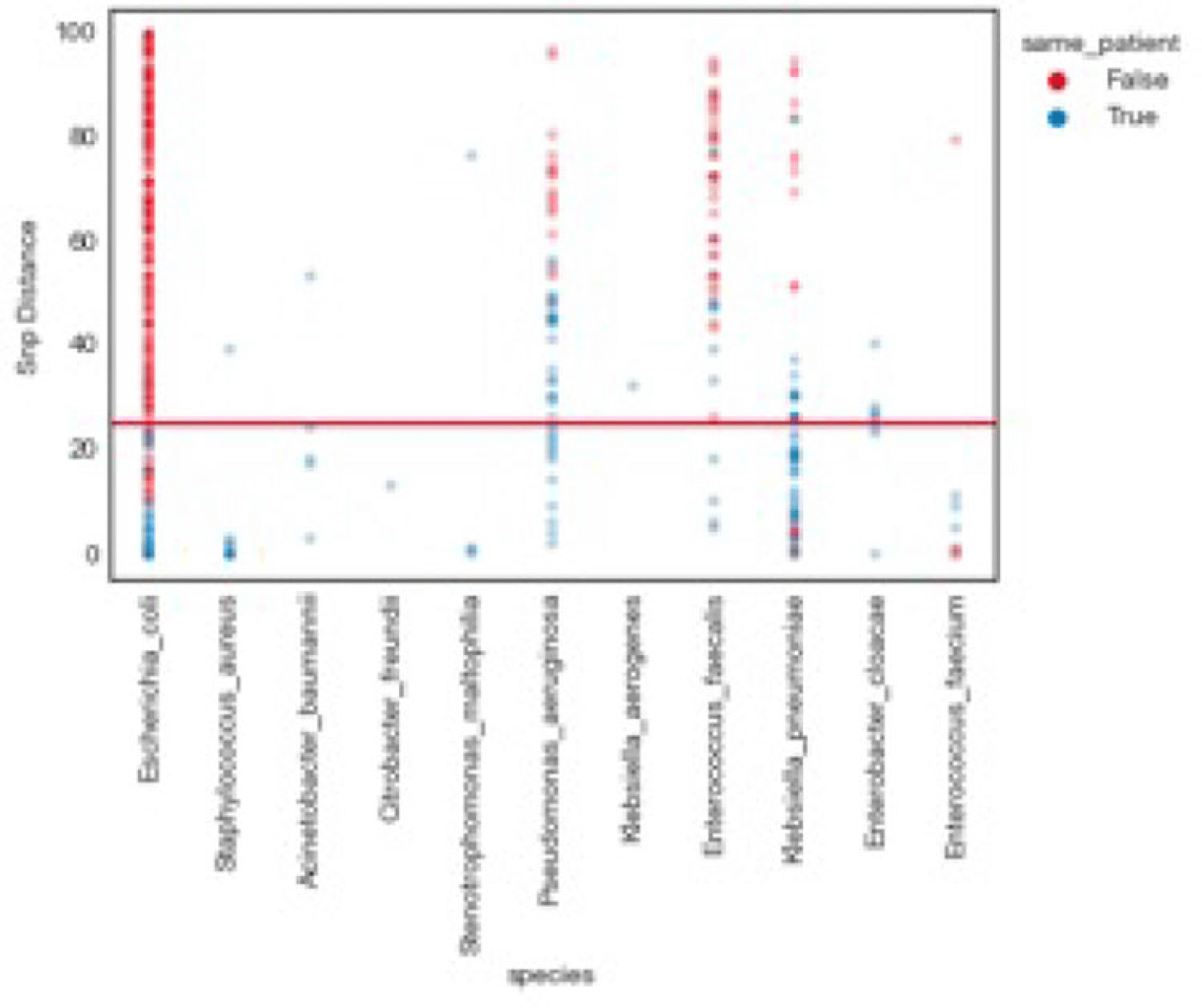
Background: Traditional hospital outbreak-detection methods are typically limited to select multidrug-resistant pathogens in a single unit, which can miss transmission of many medically important healthcare-transmissible pathogens. Whole-genome sequencing (WGS) enables comprehensive genomic resolution for accurate identification of clonal transmission. Previously, lack of scalability limited the use of WGS for hospital surveillance. Methods: We conducted prospective surveillance of select bacteria from all inpatient clinical cultures plus all bacteria from clinical cultures from ICUs and oncology units at the University of California Irvine (UCI) Clinical Microbiology Laboratory from September 2021 to February 2022. Due to pandemic stressors, this pilot test was a prelude to a real-time demonstration project. Its goal was to demonstrate the efficiency and scalability of the WGS platform when receiving samples monthly and analyzing results quarterly without the intent for real-time response. Bacterial isolates slated for discard were collected weekly and sent monthly to Day Zero Diagnostics for sequencing. In total, 1,036 samples from 926 patients were analyzed for genomic relatedness, a scalable and automated analysis pipeline already in use for rapid (days) characterization of genomic-relatedness in small and large sets of isolates. Mapping and SNP calling was performed against high-quality, best-match reference genomes. Sets of samples with pairwise distance of 2 persons with genomically related isolates and were denoted as “clusters.” Separately, we also investigated within-patient diversity by quantifying the genomic relatedness of isolates collected from individual patients. Results: Isolates represented 28 distinct species. We identified 10 Escherichia coli clusters (range, 2–4 patients; median, 2 patients), 2 Klebsiella pneumoniae clusters (range, 2–4 patients), and 1 Enterococcus faecium cluster (3 patients). All but 1 involved genomically matched isolates from multiple hospital locations. There were 4 Escherichia coli ST131 clusters spanning 4 months, including 1 with 4 patients across 3 different hospital locations. At a species level, there were distinct differences between the observed SNP distances between samples isolated from the same versus different patients (Fig. 1). All identified clusters had not been flagged by routine outbreak detection methods used by the UCI infection prevention program. Conclusions: Comprehensive WGS-based surveillance of hospital clinical isolates identified multiple potential transmission events between patients not in the same unit at the time cultures were taken. Combining WGS detection and real-time epidemiologic investigation may identify new avenues of transmission risk and could provide early warnings of clonal transmission to prevent larger outbreaks. High-volume surveillance of hospital isolates can also provide species- and context-specific clonality.
Financial support: This study was funded by Day Zero Diagnostics.
Disclosures: None
Multicenter evaluation of contamination of the healthcare environment near patients with Candida auris skin colonization – ERRATUM
- Sarah E. Sansom, Gabrielle M. Gussin, Raveena D Singh, Pamela B Bell, Ellen Benson, Jinal Makhija, Mary Carl Froilan, Raheeb Saavedra, Robert Pedroza, Christine Thotapalli, Christine Fukuda, Ellen Gough, Stefania Marron, Maria Del Mar Villanueva Guzman, Julie A. Shimabukuro, Lydia Mikhail, Stephanie Black, Massimo Pacilli, Hira Adil, Cassiana E. Bittencourt, Matthew Zahn, Nicholas Moore, D. Sexton, Judith Noble-Wang, Meghan Lyman, Michael Lin, Susan Huang, Mary K. Hayden
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 07 October 2022, e166
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
Multicenter evaluation of contamination of the healthcare environment near patients with Candida auris skin colonization
- Sarah Sansom, Gabrielle M. Gussin, Raveena D Singh, Pamela B Bell, Ellen Benson Jinal, Makhija, Raheeb Froilan, Raheeb Saavedra, Robert Pedroza, Christine Thotapalli, Christine Fukuda, Ellen Gough, Stefania Marron, Maria Del Mar Villanueva Guzman, Julie A. Shimabukuro, Lydia Mikhail, Stephanie Black, Massimo Pacilli, Hira Adil, Cassiana E. Bittencourt, Matthew Zahn, Nicholas Moore, D. Joseph Sexton, Judith Noble-Wang, Meghan Lyman, Michael Lin, Susan Huang, Mary Hayden
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s78-s79
-
- Article
-
- You have access Access
- Open access
- Export citation
-
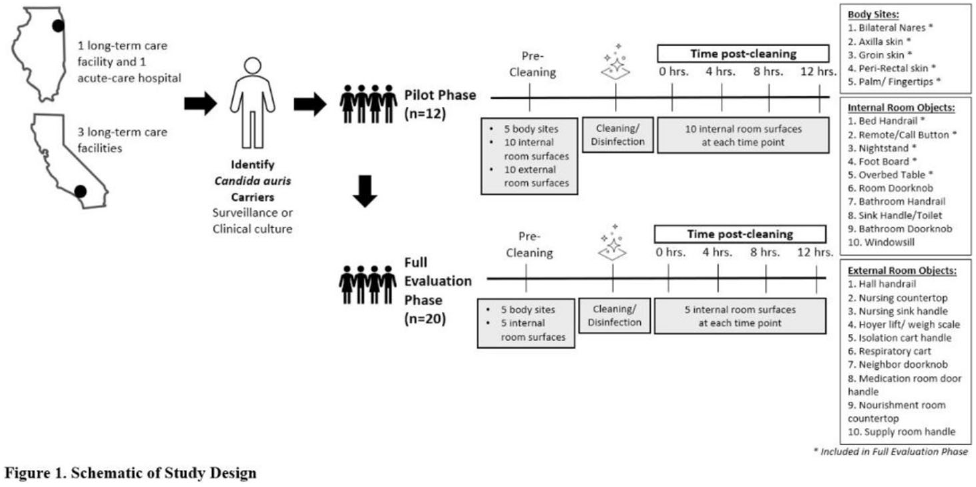
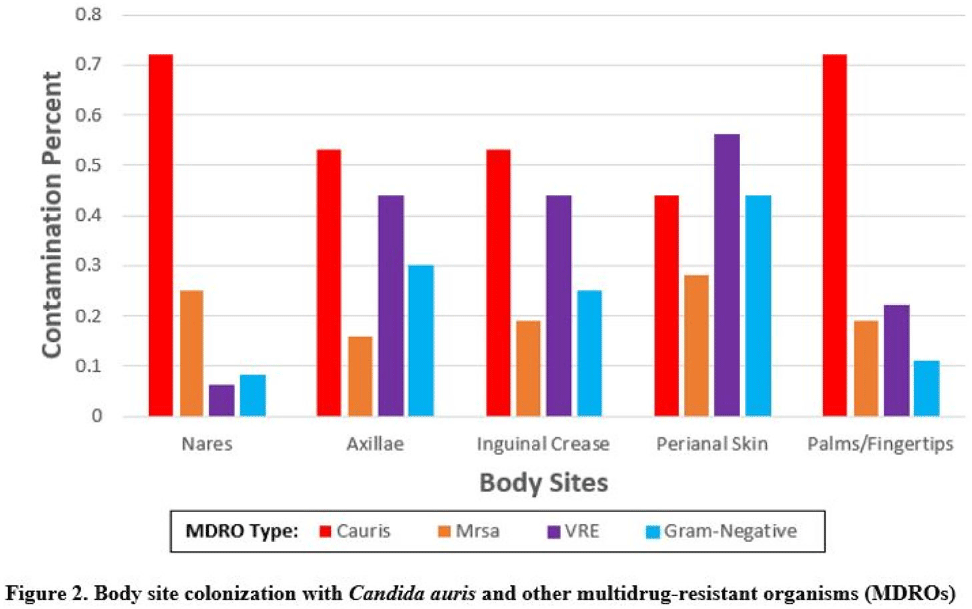
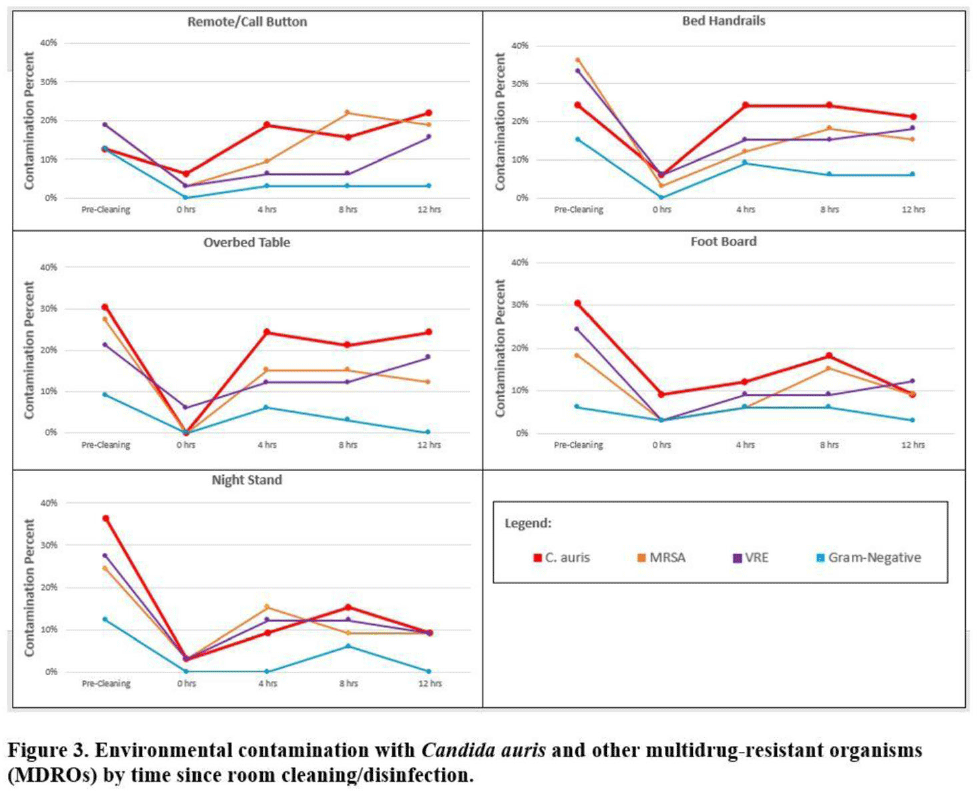
Background:Candida auris is an emerging multidrug-resistant yeast that is transmitted in healthcare facilities and is associated with substantial morbidity and mortality. Environmental contamination is suspected to play an important role in transmission but additional information is needed to inform environmental cleaning recommendations to prevent spread. Methods: We conducted a multiregional (Chicago, IL; Irvine, CA) prospective study of environmental contamination associated with C. auris colonization of patients and residents of 4 long-term care facilities and 1 acute-care hospital. Participants were identified by screening or clinical cultures. Samples were collected from participants’ body sites (eg, nares, axillae, inguinal creases, palms and fingertips, and perianal skin) and their environment before room cleaning. Daily room cleaning and disinfection by facility environmental service workers was followed by targeted cleaning of high-touch surfaces by research staff using hydrogen peroxide wipes (see EPA-approved product for C. auris, List P). Samples were collected immediately after cleaning from high-touch surfaces and repeated at 4-hour intervals up to 12 hours. A pilot phase (n = 12 patients) was conducted to identify the value of testing specific high-touch surfaces to assess environmental contamination. High-yield surfaces were included in the full evaluation phase (n = 20 patients) (Fig. 1). Samples were submitted for semiquantitative culture of C. auris and other multidrug-resistant organisms (MDROs) including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus (VRE), extended-spectrum β-lactamase–producing Enterobacterales (ESBLs), and carbapenem-resistant Enterobacterales (CRE). Times to room surface contamination with C. auris and other MDROs after effective cleaning were analyzed. Results:Candida auris colonization was most frequently detected in the nares (72%) and palms and fingertips (72%). Cocolonization of body sites with other MDROs was common (Fig. 2). Surfaces located close to the patient were commonly recontaminated with C. auris by 4 hours after cleaning, including the overbed table (24%), bed handrail (24%), and TV remote or call button (19%). Environmental cocontamination was more common with resistant gram-positive organisms (MRSA and, VRE) than resistant gram-negative organisms (Fig. 3). C. auris was rarely detected on surfaces located outside a patient’s room (1 of 120 swabs; <1%). Conclusions: Environmental surfaces near C. auris–colonized patients were rapidly recontaminated after cleaning and disinfection. Cocolonization of skin and environment with other MDROs was common, with resistant gram-positive organisms predominating over gram-negative organisms on environmental surfaces. Limitations include lack of organism sequencing or typing to confirm environmental contamination was from the room resident. Rapid recontamination of environmental surfaces after manual cleaning and disinfection suggests that alternate mitigation strategies should be evaluated.
Funding: None
Disclosures: None
Impact of Roommates on MDRO Spread in Nursing Homes
- Gabrielle M. Gussin, Ken Kleinman, Raveena D. Singh, Raheeb Saavedra, Lauren Heim, Marlene Estevez, Tabitha D. Catuna, Eunjung Lee, Avy Osalvo, Kaye D. Evans, Julie A. Shimabukuro, James A. McKinnell, Loren Miller, Cassiana E. Bittencourt, Ellena M. Peterson, Susan Huang
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s66-s67
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Addressing the high burden of multidrug-resistant organisms (MDROs) in nursing homes is a public health priority. High interfacility transmission may be attributed to inadequate infection prevention practices, shared living spaces, and frequent care needs. We assessed the contribution of roommates to the likelihood of MDRO carriage in nursing homes. Methods: We performed a secondary analysis of the SHIELD OC (Shared Healthcare Intervention to Eliminate Life-threatening Dissemination of MDROs in Orange County, CA) Project, a CDC-funded regional decolonization intervention to reduce MDROs among 38 regional facilities (18 nursing homes, 3 long-term acute-care hospitals, and 17 hospitals). Decolonization in participating nursing homes involved routine chlorhexidine bathing plus nasal iodophor (Monday through Friday, twice daily every other week) from April 2017 through July 2019. MDRO point-prevalence assessments involving all residents at 16 nursing homes conducted at the end of the intervention period were used to determine whether having a roommate was associated with MDRO carriage. Nares, bilateral axilla/groin, and perirectal swabs were processed for methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant enterococcus (VRE), extended-spectrum β-lactamase (ESBL)–producing Enterobacteriaceae, and carbapenem-resistant Enterobacteriaceae (CRE). Generalized linear mixed models assessed the impact of maximum room occupancy on MDRO prevalence when clustering by room and hallway, and adjusting for the following factors: nursing home facility, age, gender, length-of-stay at time of swabbing, bedbound status, known MDRO history, and presence of urinary or gastrointestinal devices. CRE models were not run due to low counts. Results: During the intervention phase, 1,451 residents were sampled across 16 nursing homes. Overall MDRO prevalence was 49%. In multivariable models, we detected a significant increasing association of maximum room occupants and MDRO carriage for MRSA but not other MDROs. For MRSA, the adjusted odds ratios for quadruple-, triple-, and double-occupancy rooms were 3.5, 3.6, and 2.8, respectively, compared to residents in single rooms (P = .013). For VRE, these adjusted odds ratios were 0.3, 0.3, and 0.4, respectively, compared to residents in single rooms (P = NS). For ESBL, the adjusted odds ratios were 0.9, 1.1, and 1.5, respectively, compared to residents in single rooms (P = nonsignificant). Conclusions: Nursing home residents in shared rooms were more likely to harbor MRSA, suggesting MRSA transmission between roommates. Although decolonization was previously shown to reduce MDRO prevalence by 22% in SHIELD nursing homes, this strategy did not appear to prevent all MRSA transmission between roommates. Additional efforts involving high adherence hand hygiene, environmental cleaning, and judicious use of contact precautions are likely needed to reduce transmission between roommates in nursing homes.
Funding: None
Disclosures: Gabrielle M. Gussin, Stryker (Sage Products): Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Clorox: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Medline: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes. Xttrium: Conducting studies in which contributed antiseptic product is provided to participating hospitals and nursing homes.







