6 results
Multicenter evaluation of contamination of the healthcare environment near patients with Candida auris skin colonization – ERRATUM
- Sarah E. Sansom, Gabrielle M. Gussin, Raveena D Singh, Pamela B Bell, Ellen Benson, Jinal Makhija, Mary Carl Froilan, Raheeb Saavedra, Robert Pedroza, Christine Thotapalli, Christine Fukuda, Ellen Gough, Stefania Marron, Maria Del Mar Villanueva Guzman, Julie A. Shimabukuro, Lydia Mikhail, Stephanie Black, Massimo Pacilli, Hira Adil, Cassiana E. Bittencourt, Matthew Zahn, Nicholas Moore, D. Sexton, Judith Noble-Wang, Meghan Lyman, Michael Lin, Susan Huang, Mary K. Hayden
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue 1 / 2022
- Published online by Cambridge University Press:
- 07 October 2022, e166
-
- Article
-
- You have access Access
- Open access
- HTML
- Export citation
Outbreak investigation of CRAB at an acute-care hospital ICU during the COVID-19 pandemic–Chicago, Illinois, March 2020–September 2021
- Hira Adil, Kelly Walblay, Shelby Daniel-Wayman, Massimo Pacilli, Shannon Xydis, Christine Pate, Ann Valley, Stephanie Black
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, p. s84
-
- Article
-
- You have access Access
- Open access
- Export citation
-
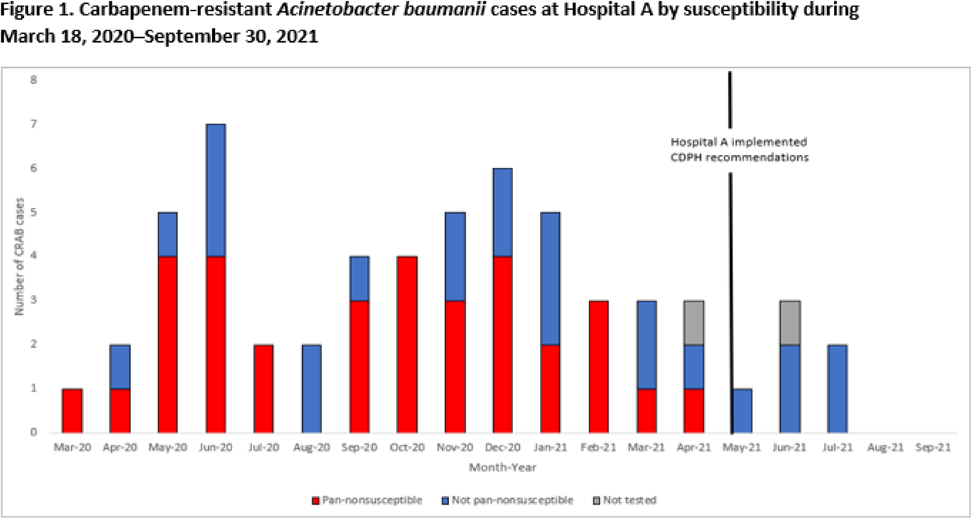
Background: Carbapenem-resistant Acinetobacter baumannii (CRAB) is primarily associated with hospital-acquired infections and is an urgent public health threat due to its ability to contaminate the environment and cause severe disease. In 2019, Illinois began pilot surveillance for CRAB requiring select laboratories to submit specimens for molecular characterization. On July 17, 2020, the Chicago Department of Public Health (CDPH) was notified of an increase in CRAB infections in a 20-bed ICU at an acute-care hospital in Chicago (hospital A) during the initial COVID-19 surge. We summarize the outbreak investigation findings and infection control recommendations. Methods: Clinical cultures were collected from patients in hospital A, and CRAB-positive isolates were sent to the Wisconsin State Laboratory of Hygiene for mechanism of resistance and antibiotic susceptibility testing. On-site assessments and remote follow-ups were conducted by CDPH infection preventionists to evaluate infection control practices including environmental cleaning, hand hygiene compliance, and use of personal protective equipment (PPE). The Illinois Department of Public Health and CDPH summarized the testing results, facilitated a containment response, and provided recommendations for infection control. Results: From March 18, 2020, to September 30, 2021, 56 patients with CRAB infections were identified from hospital A, and 33 (59%) of these cases were pan-nonsusceptible. Most specimen sources were sputum (n = 30, 54%), followed by blood (n = 13, 23%), urine (n = 6, 11%) and other (n = 7, 13%). Among isolates with mechanism testing (n = 54), 45 (83%) were positive for OXA-24/40 and 9 (17%) were positive for OXA-23. Of the CRAB-positive patients, 28 (50%) were previously positive for SARS-CoV-2. To date, 25 of these patients (45%) have been discharged and 31 (55%) have died. Two onsite visits and 7 remote-assistance sessions were conducted as part of the investigation. In response to increased COVID-19 hospitalizations, hospital A moved to crisis-capacity PPE use and encountered staffing shortages, which led to compromised infection control measures. Cleaning agents (Quat disinfectant cleaner) were also found to be ineffective against CRAB and required long contact times. Conclusions: In response to the CRAB outbreak at hospital A, CDPH recommended that the hospital stop crisis-capacity protocols for PPE, conduct admission screening and point-prevalence testing for CRAB, implement a hand hygiene campaign, and use an EPA-registered List K product for environmental cleaning. These recommendations were implemented in May 2021, and no CRAB cases have been reported since July 2021. To reduce CRAB transmission during the pandemic, facility leadership must commit resources to educate staff on effective infection control practices including conventional use of PPE, appropriate cleaning agents, and improved hand hygiene.
Funding: None
Disclosures: None
Multicenter evaluation of contamination of the healthcare environment near patients with Candida auris skin colonization
- Sarah Sansom, Gabrielle M. Gussin, Raveena D Singh, Pamela B Bell, Ellen Benson Jinal, Makhija, Raheeb Froilan, Raheeb Saavedra, Robert Pedroza, Christine Thotapalli, Christine Fukuda, Ellen Gough, Stefania Marron, Maria Del Mar Villanueva Guzman, Julie A. Shimabukuro, Lydia Mikhail, Stephanie Black, Massimo Pacilli, Hira Adil, Cassiana E. Bittencourt, Matthew Zahn, Nicholas Moore, D. Joseph Sexton, Judith Noble-Wang, Meghan Lyman, Michael Lin, Susan Huang, Mary Hayden
-
- Journal:
- Antimicrobial Stewardship & Healthcare Epidemiology / Volume 2 / Issue S1 / July 2022
- Published online by Cambridge University Press:
- 16 May 2022, pp. s78-s79
-
- Article
-
- You have access Access
- Open access
- Export citation
-
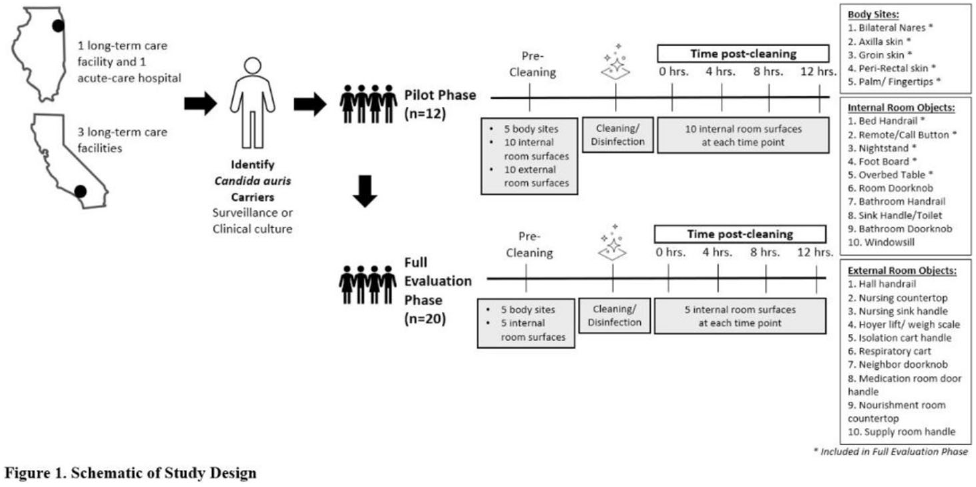
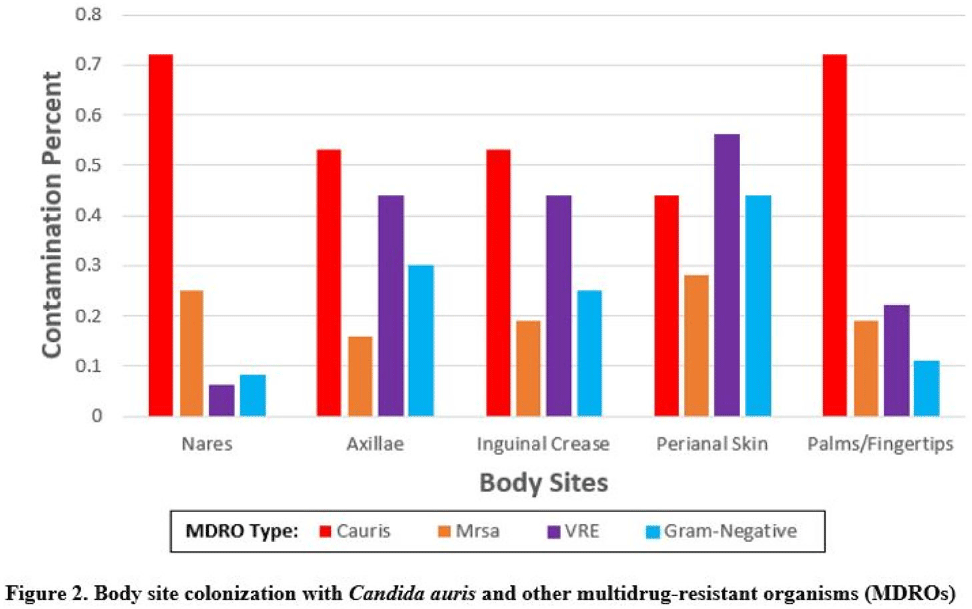
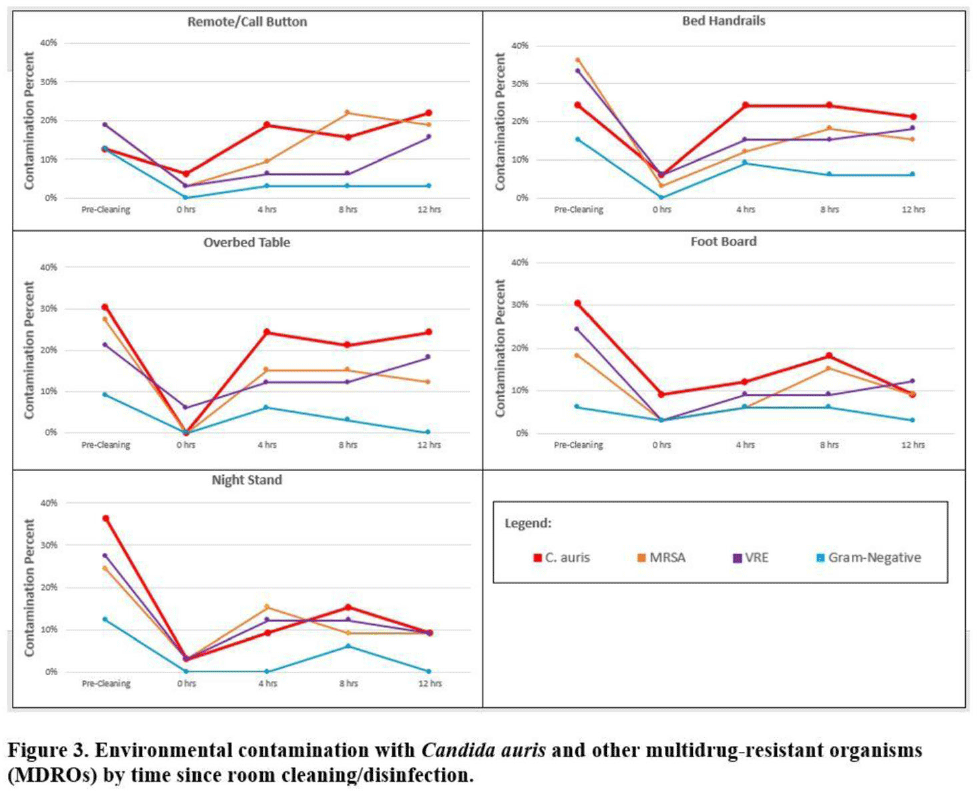
Background:Candida auris is an emerging multidrug-resistant yeast that is transmitted in healthcare facilities and is associated with substantial morbidity and mortality. Environmental contamination is suspected to play an important role in transmission but additional information is needed to inform environmental cleaning recommendations to prevent spread. Methods: We conducted a multiregional (Chicago, IL; Irvine, CA) prospective study of environmental contamination associated with C. auris colonization of patients and residents of 4 long-term care facilities and 1 acute-care hospital. Participants were identified by screening or clinical cultures. Samples were collected from participants’ body sites (eg, nares, axillae, inguinal creases, palms and fingertips, and perianal skin) and their environment before room cleaning. Daily room cleaning and disinfection by facility environmental service workers was followed by targeted cleaning of high-touch surfaces by research staff using hydrogen peroxide wipes (see EPA-approved product for C. auris, List P). Samples were collected immediately after cleaning from high-touch surfaces and repeated at 4-hour intervals up to 12 hours. A pilot phase (n = 12 patients) was conducted to identify the value of testing specific high-touch surfaces to assess environmental contamination. High-yield surfaces were included in the full evaluation phase (n = 20 patients) (Fig. 1). Samples were submitted for semiquantitative culture of C. auris and other multidrug-resistant organisms (MDROs) including methicillin-resistant Staphylococcus aureus (MRSA), vancomycin-resistant Enterococcus (VRE), extended-spectrum β-lactamase–producing Enterobacterales (ESBLs), and carbapenem-resistant Enterobacterales (CRE). Times to room surface contamination with C. auris and other MDROs after effective cleaning were analyzed. Results:Candida auris colonization was most frequently detected in the nares (72%) and palms and fingertips (72%). Cocolonization of body sites with other MDROs was common (Fig. 2). Surfaces located close to the patient were commonly recontaminated with C. auris by 4 hours after cleaning, including the overbed table (24%), bed handrail (24%), and TV remote or call button (19%). Environmental cocontamination was more common with resistant gram-positive organisms (MRSA and, VRE) than resistant gram-negative organisms (Fig. 3). C. auris was rarely detected on surfaces located outside a patient’s room (1 of 120 swabs; <1%). Conclusions: Environmental surfaces near C. auris–colonized patients were rapidly recontaminated after cleaning and disinfection. Cocolonization of skin and environment with other MDROs was common, with resistant gram-positive organisms predominating over gram-negative organisms on environmental surfaces. Limitations include lack of organism sequencing or typing to confirm environmental contamination was from the room resident. Rapid recontamination of environmental surfaces after manual cleaning and disinfection suggests that alternate mitigation strategies should be evaluated.
Funding: None
Disclosures: None
Regional Impact of a CRE Intervention Targeting High Risk Postacute Care Facilities (Chicago PROTECT)
- Michael Lin, Mary Carl Froilan, Jinal Makhija, Ellen Benson, Sarah Bartsch, Pamela B. Bell, Stephanie Black, Deborah Burdsall, Michelle Ealy, Anthony Fiore, Sharon Foy, Mabel Frias, Alice Han, David Hines, Olufemi Jegede, John Jernigan, Sarah K. Kemble, Mary Alice Lavin, Bruce Lee, George Markovski, Massimo Pacilli, Sujan Reddy, Erica Runningdeer, Michael Schoeny, Mitali Shah, Rachel Slayton, Elizabeth Soda, Nimalie Stone, Angela S. Tang, Karen Trimberger, Marion Tseng, Yingxu Xiang, Robert Weinstein, William Trick, Mary Hayden
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s48-s49
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Carbapenem-resistant Enterobacteriaceae (CRE) are endemic in the Chicago region. We assessed the regional impact of a CRE control intervention targeting high-prevalence facilities; that is, long-term acute-care hospitals (LTACHs) and ventilator-capable skilled nursing facilities (vSNFs). Methods: In July 2017, an academic–public health partnership launched a regional CRE prevention bundle: (1) identifying patient CRE status by querying Illinois’ XDRO registry and periodic point-prevalence surveys reported to public health, (2) cohorting or private rooms with contact precautions for CRE patients, (3) combining hand hygiene adherence, monitoring with general infection control education, and guidance by project coordinators and public health, and (4) daily chlorhexidine gluconate (CHG) bathing. Informed by epidemiology and modeling, we targeted LTACHs and vSNFs in a 13-mile radius from the coordinating center. Illinois mandates CRE reporting to the XDRO registry, which can also be manually queried or generate automated alerts to facilitate interfacility communication. The regional intervention promoted increased automation of alerts to hospitals. The prespecified primary outcome was incident clinical CRE culture reported to the XDRO registry in Cook County by month, analyzed by segmented regression modeling. A secondary outcome was colonization prevalence measured by serial point-prevalence surveys for carbapenemase-producing organism colonization in LTACHs and vSNFs. Results: All eligible LTACHs (n = 6) and vSNFs (n = 9) participated in the intervention. One vSNF declined CHG bathing. vSNFs that implemented CHG bathing typically bathed residents 2–3 times per week instead of daily. Overall, there were significant gaps in infection control practices, especially in vSNFs. Also, 75 Illinois hospitals adopted automated alerts (56 during the intervention period). Mean CRE incidence in Cook County decreased from 59.0 cases per month during baseline to 40.6 cases per month during intervention (P < .001). In a segmented regression model, there was an average reduction of 10.56 cases per month during the 24-month intervention period (P = .02) (Fig. 1), and an estimated 253 incident CRE cases were averted. Mean CRE incidence also decreased among the stratum of vSNF/LTACH intervention facilities (P = .03). However, evidence of ongoing CRE transmission, particularly in vSNFs, persisted, and CRE colonization prevalence remained high at intervention facilities (Table 1). Conclusions: A resource-intensive public health regional CRE intervention was implemented that included enhanced interfacility communication and targeted infection prevention. There was a significant decline in incident CRE clinical cases in Cook County, despite high persistent CRE colonization prevalence in intervention facilities. vSNFs, where understaffing or underresourcing were common and lengths of stay range from months to years, had a major prevalence challenge, underscoring the need for aggressive infection control improvements in these facilities.
Funding: The Centers for Disease Control and Prevention (SHEPheRD Contract No. 200-2011-42037)
Disclosures: M.Y.L. has received research support in the form of contributed product from OpGen and Sage Products (now part of Stryker Corporation), and has received an investigator-initiated grant from CareFusion Foundation (now part of BD).
Repeated Prevalence Surveys and Admission Screening for Candida auris at One Long-Term Acute-Care Hospital, Chicago, 2016–2019
- Massimo Pacilli, Kelly Walblay, Hira Adil, Shannon Xydis, Janna Kerins, Ann Valley, Stephanie Black
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s14-s15
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background: Since the initial identification of Candida auris in 2016 in Chicago, ongoing spread has been documented in the Chicago area, primarily among older adults with complex medical issues admitted to high-acuity long-term care facilities, including long-term acute-care hospitals (LTACHs). As of October 2019, 790 cases have been reported in Illinois. Knowing C. auris colonization status on admission is important for prompt implementation of infection control precautions. We describe periodic facility point-prevalence surveys (PPSs) and admission screening at LTACH A. Methods: Beginning September 2016, we conducted repeated PPSs for C. auris colonization at LTACH A. After a baseline PPS, we initiated admission screening in May 2019 for patients without prior evidence of C. auris colonization or infection. C. auris screening specimens consisted of composite bilateral axillary/inguinal swabs tested at public health laboratories. We compared a limited set of patient characteristics based on admission screening results. Results: From September 2016 through October 2019, 277 unique patients were screened at LTACH A during 10 PPSs. Overall, 36 patients (13%) were identified to be colonized. The median facility C. auris prevalence increased from 2.8% in 2016 to 37% in 2019 (Fig. 1). During May–September 2019, among 174 unique patients admitted, 151 (87%) were screened for C. auris colonization on admission, of whom 18 (12%) were found to be colonized. Overall, 14 patients were known to have C. auris colonization on admission and were not rescreened, and 9 patients were discharged before screening specimens could be collected. A significantly higher proportion of patients testing positive for C. auris on admission had a central venous catheter or a peripherally inserted central catheter or were already on contact precautions (Table 1). The PPS conducted on October 1, 2019, revealed 5 new C. auris colonized patients who had screened negative on admission. Conclusions: Repeated PPSs at LTACH A indicated control of C. auris transmission in 2016–2017, followed by increasing prevalence beginning in May 2018, likely from patients admitted with unrecognized C. auris colonization and subsequent facility spread. Admission screening allowed for early detection of C. auris colonization. However, identification during subsequent PPS of additional colonized patients indicates that facility transmission is ongoing. Both admission screening and periodic PPSs are needed for timely detection of colonized patients. Given the high C. auris prevalence in LTACHs and challenges in identifying readily apparent differences between C. auris positive and negative patients on admission, we recommend that all patients being admitted to an LTACH in endemic areas should be screened for C. auris.
Funding: None
Disclosures: None
Candida auris and Carbapenemase-Producing Organism Prevalence in an Extended Stay Pediatric Hospital, Chicago, Illinois, 2019
- Kelly Walblay, Tristan McPherson, Elissa Roop, David Soglin, Ann Valley, Latania Logan, Snigdha Vallabhaneni, Stephanie Black, Massimo Pacilli
-
- Journal:
- Infection Control & Hospital Epidemiology / Volume 41 / Issue S1 / October 2020
- Published online by Cambridge University Press:
- 02 November 2020, pp. s145-s146
- Print publication:
- October 2020
-
- Article
-
- You have access Access
- Export citation
-
Background:Candida auris and carbapenemase-producing organisms (CPO) are multidrug-resistant organisms that can colonize people for prolonged periods and can cause invasive infections and spread in healthcare settings, particularly in high-acuity long-term care facilities. Point-prevalence surveys (PPSs) conducted in long-term acute-care hospitals in the Chicago region identified median prevalence of colonization to be 31% for C. auris and 24% for CPO. Prevalence of C. auris colonization has not been described in pediatric populations in the United States, and limited data exist on CPO colonization in children outside intensive care units. The Chicago Department of Public Health (CDPH) conducted a PPS to assess C. auris and CPO colonization in a pediatric hospital serving high-acuity patients with extended lengths of stay (LOS). Methods: CDPH conducted a PPS in August 2019 in a pediatric hospital with extended LOS to screen for C. auris and CPO colonization. Medical devices (ie, gastrostomy tubes, tracheostomies, mechanical ventilators, and central venous catheters [CVC]) and LOS were documented. Screening specimens consisted of composite bilateral axillae and groin swabs for C. auris and rectal swabs for CPO testing. The Wisconsin State Laboratory of Hygiene tested all specimens. Real-time polymerase chain reaction (PCR) assays were used to detect C. auris DNA and carbapenemase genes: blaKPC, blaNDM, blaVIM, blaOXA-48, and blaIMP (Xpert Carba-R Assay, Cepheid, Sunnyvale, CA). All axillae and groin swabs were processed by PCR and culture to identify C. auris. For CPO, culture was only performed on PCR-positive specimens. Results: Of the 29 patients hospitalized, 26 (90%) had gastrostomy tubes, 24 (83%) had tracheostomies, 20 (69%) required mechanical ventilation, and 3 (10%) had CVCs. Also, 25 (86%) were screened for C. auris and CPO; 4 (14%) lacked parental consent and were not swabbed. Two rectal specimens were unsatisfactory, producing invalid CPO test results. Median LOS was 35 days (range, 1–300 days). No patients were positive for C. auris. From CPO screening, blaOXA-48 was detected in 1 patient sample, yielding a CPO prevalence of 3.4% (1 of 29). No organism was recovered from the blaOXA-48 positive specimen. Conclusions: This is the first documented screening of C. auris colonization in a pediatric hospital with extended LOS. Despite a high prevalence of C. auris and CPOs in adult healthcare settings of similar acuity in the region, C. auris was not identified and CPOs were rare at this pediatric facility. Additional evaluations in pediatric hospitals should be conducted to further understand C. auris and CPO prevalence in this population.
Funding: None
Disclosures: None







