Volume 54 - October 2006
Research Article
Mineralogy and Geochemistry of the Host-Rock Alterations Associated with the Shea Creek Unconformity-Type Uranium Deposits (Athabasca Basin, Saskatchewan, Canada). Part 1. Spatial Variation of Illite Properties
- Emmanuel Laverret, Patricia Patrier Mas, Daniel Beaufort, Philippe Kister, David Quirt, Patrice Bruneton, Norbert Clauer
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 275-294
-
- Article
- Export citation
-
Unconformity-related uranium deposits, which represent a significant high-grade uranium resource, are systematically surrounded by a host-rock alteration halo enriched in clay minerals. Illite is often the major clay mineral component of the halo and it displays a variable crystal structure. New data are provided on the crystal structure and the chemistry of illite encountered within and outside of the alteration halo surrounding the Shea Creek deposit. Two illite populations were distinguished using textural and structural criteria: samples rich in the tv-1M polytype display thin (sub-micrometer) and ‘hairy’ shapes, while samples richer in the cv-1M polytype contain illites with rigid lath-like shapes several micrometers wide. In barren ‘regional’ sandstone, the trends with depth of the textural and microstructural properties of illite particles are: (1) an increase of particle size, (2) an evolution to a more isometric form, and (3) a dominance of the cv-1M polytype over the tv-1M polytype. These trends record diagenetic processes under conditions of deep burial and differ from those observed in altered sandstone around the uranium mineralization. The altered sandstone is characterized by enrichment in the tv-1M polytype near the unconformity and/or brittle structural features. This tv-1M illitization took place in response to structurally-controlled infiltration of basement rocks by diagenetic brines which were further recycled after interaction into the overlying basin. Variations of the illite structural and textural properties may result from nucleation/growth kinetics and may be indicative of a change in the flow regime, and/or a change of saturation state of the fluid vs. illite. The tv-1M illite may be favored in environments characterized by a high fluid/rock ratio and a high supersaturation state of the fluids in proximity to mineralization.
A New Ni-Rich Stevensite From the Ophiolite Complex of Othrys, Central Greece
- George E. Christidis, Ioannis Mitsis
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 653-666
-
- Article
- Export citation
-
The first occurrence of Ni-rich stevensite found in the ophiolite complex of Othrys, Central Greece is described. The stevensite, which develops in cracks in a host serpentinite, formed at the expense of serpentine. Two varieties of stevensite have been described: a Mg-rich, Ni-poorer variety with 0.4–1.2 octahedral Ni atoms per half formula unit (p.h.f.u.) and a Ni-rich variety with >2 Ni atoms p.h.f.u. The layer charge in both varieties is −0.24 p.h.f.u.. Stevensite layers are completely separated when dispersed in dilute polyvinylpyrrolidone (PVP) solutions and begin to convert to talc after heating at 250°C for 90 min. Total conversion to talc is observed at 550°C. Formation of Ni-rich stevensite took place at ambient temperature during supergene processes. The scarcity of Ni-rich stevensite occurrences in nature is attributed to the metastability of smectite and to the analytical procedures used in previous studies. Stevensite is considered a phase containing domains with variable numbers of octahedral vacancies. A new experimental protocol is proposed for the determination of Ni-rich stevensite, based on a combination of XRD after solvation with various organic liquids and subsequent heating at 750°C.
FTIR investigation of the evolution of the octahedral sheet of kaolinite-smectite with progressive kaolinization
- Javier Cuadros, Teresa Dudek
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 1-11
-
- Article
- Export citation
-
Twenty two samples were studied to investigate the nature and evolution mechanism of mixed-layer kaolinite-smectite (K-S). We examined the <2 µm or <0.2 µm fraction of K-S formed by hydrothermal and hypergenic alteration of volcanic material. The samples are from three localities: 20 specimens from a Tortonian clay deposit in Almería, Spain; one specimen from weathered Eocene volcanic ash from the Yucatan Peninsula, Mexico; and one sample from a weathered Jurassic bentonite from Northamptonshire, England. The samples were studied using chemical analysis, X-ray diffraction (XRD) and Fourier transform infrared spectroscopy (FTIR). The XRD patterns of the oriented, glycolated mounts were modeled using NEWMOD and the proportion of smectite and kaolinite layers was determined, ranging between 0 and 80% kaolinite. The analysis of the OH-stretching region of the FTIR spectra at different temperatures (180–550°C) showed the progressive dehydroxylation of kaolinite domains and, perhaps, of smectite domains, but no detailed information could be obtained about the sequential OH loss in different cation environments. The abundance and short-range ordering of the octahedral cations were studied using the OH-bending bands. The chemical and FTIR-estimated octahedral cation abundances were broadly similar. Aluminum showed a tendency to mix with Fe and Mg rather than to form AlAl pairs. Al-for-Mg substitution accompanying kaolinization was evident from the increase in AlAl pairs and decrease in AlMg pairs. Iron is retained in the structure. No other octahedral cation rearrangement was observed. The intensity of the 750 cm−1 band, assigned to translational vibrations of external OH groups in a kaolinitic environment, was quantified and modeled in relation to kaolinite layer proportion. The chemical data show that there are residual interlayer cations in kaolinite domains, which, in accordance with the model mentioned above, disturb external OH-translation vibrations. These results indicate the persistence of certain chemical and structural smectite features in kaolinite domains and thus support a smectite kaolinization process via a solid-state transformation. This confirms previous XRD, thermal, chemical and NMR analyses of the same sample set.
Crystal-Size Dependence of Illite-Smectite Isotope Equilibration with Changing Fluids
- Lynda B. Williams, Richard L. Hervig
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 531-540
-
- Article
- Export citation
-
Differences in equilibration rates among crystals of different sizes may be used to deduce paleofluid changes over time if the crystal-growth mechanism is known. To explore isotopic equilibration rates as a function of illite growth, we studied B-isotope changes during illitization of smectite. Montmorillonite (<2.0 µm SWy-1, K saturated) was reacted with aqueous boric acid (1000 ppm B) at 300°C, 100 MPa in sealed Au capsules (1:1 fluid:mineral ratio). The initial fluid was 0‰ (NBS 951 standard) but after R1 ordering occurred (65 days of reaction) the fluid was changed to −7‰ in order to examine the rate of isotopic re-equilibration. Samples were taken intermittently throughout the experiment. Each aliquot was NH4 exchanged and size separated into fine (<0.2 µm), medium (0.2–2.0 µm) and coarse (>2.0 µm) fractions. The isotopic composition of B in the tetrahedral sheet was then measured for comparison with the predicted equilibrium values.
The fine fraction showed equilibrium isotope ratios within 10 days, indicating that small, newly nucleated crystals precipitate in equilibrium with the fluid under supersaturated, closed conditions. These fine-fraction minerals did not re-equilibrate when the fluid was changed. The medium fraction gradually equilibrated with the initial fluid as illite grew to values >50%, but did not re-equilibrate with the later fluid. The coarse fraction was slow to begin recrystallization, perhaps due to dissolution kinetics of large crystals or the presence of detrital contaminants. However, it showed the fastest rate of isotopic change with crystal growth after R1 ordering. We conclude that at 300°C, the initial B–O bonds formed in illite are stable, and isotopic re-equilibration only occurs on new crystal growth. Therefore, different isotope ratios are preserved in different crystal size fractions due to different rates of crystal growth. Large crystals may reflect equilibrium with recent fluid while smaller crystals may retain isotope compositions reflecting equilibrium with earlier fluids.
Calorimetric Determination of the Enthalpies of Formation of Hydrotalcite-Like Solids and Their Use in the Geochemical Modeling of Metals in Natural Waters
- Rama kumar Allada, Edward Peltier, Alexandra Navrotsky, William H. Casey, C. Annette Johnson, Hillary Thompson Berbeco, Donald L. Sparks
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 409-417
-
- Article
- Export citation
-
Interest in hydrotalcite-like compounds has grown due to their role in controlling the mobility of aqueous metals in the environment as well as their use as catalysts, catalyst precursors and specialty chemicals. Although these materials have been studied in a number of contexts, little is known of their thermodynamic properties. High-temperature oxide melt solution calorimetry was used to measure the standard enthalpy of formation for compounds M(II)1−xAlx(OH)2(CO3)x/2·mH2O (0.2 < x < 0.4, M(II) = Mg, Co, Ni and Zn). The enthalpy of formation of these compounds from the relevant single cation phases was also determined. The formation of HTLCs results in a 5–20 kJ/mol enthalpy stabilization from the single cation hydroxides and carbonates and water. The data are correlated to two variables: the ratio of divalent to trivalent cation in the solid (M(II)/Al) and the identity of the divalent cation. It was observed that the M(II)/Al ratio exerts a minor influence on the enthalpy of formation from single-cation phases, while greater differences in stabilization resulted from changes in the chemical nature of the divalent cation. However, the data do not support any statistically significant correlation between the composition of HTLCs and their heats of formation. Equilibrium geochemical calculations based upon the thermodynamic data illustrate the effect of HTLCs on the speciation of metals in natural waters. These calculations show that, in many cases, HTLCs form even in waters that are undersaturated with respect to the individual divalent metal hydroxides and carbonates. Phase diagrams and stability diagrams involving Ni-bearing HTLCs and the single-cation components are presented. The Ni(II) concentration as a function of pH as well as the stability diagram for the equilibrium among minerals in the CaO-NiO-Al2O3-SiO2-CO2-H2O system at 298 K are plotted.
Crystal-Chemical Factors Responsible for the Distribution of Octahedral Cations Over trans- and cis-Sites in Dioctahedral 2:1 Layer Silicates
- Victor A. Drits, Douglas K. McCarty, Bella B. Zviagina
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 131-152
-
- Article
- Export citation
-
Crystal chemical analysis of various dioctahedral 2:1 phyllosilicates consisting of trans-vacant (tv) and cis-vacant (cv) layers and interstratified cv and tv layers shows that there is compositional control over the distribution of octahedral cations over trans and cis sites. Fe3+ and Mg-rich dioctahedral micas (celadonite, glauconite, leucophyllite and most phengite) occur only as tv varieties. Similarly, the occurrence of tv illites and tv illite fundamental particles in illite-smectite (I-S) does not depend significantly on the cation composition of the 2:1 layers. In contrast, compositional restrictions exist to control the occurrence of pure cv1M illite, which can form only as Fe- and Mg-poor varieties. Similarly, proportions of cv and tv layers in illite fundamental particles depend on the amount of Al in octahedral and tetrahedral sheets of the 2:1 layers.
Simulations of atomic coordinates and interatomic distances for periodic tv1M and cv1M illite structures allow us to reveal the main structural factors that favor the formation of cv layers in illite and I-S. It is shown that in contrast to the tv1M structure, interlayer K in cv1M illite has an environment which is similar to that in 2M1 muscovite. This similarity along with a high octahedral and tetrahedral Al content probably provides stability for cv1M illite in low-temperature natural environments. Because of structural control, the occurrence of monomineral cv1M illite, its association with tv 1M illite, and interstratified cv-tv illite fundamental particles is confined by certain physical and chemical conditions. These varieties are most often formed by hydrothermal activity of different origin. The initial material for their formation should be Al-rich and the hydrothermal fluids should be Mg- and Fe-poor. They occur mostly around ore deposits, in bentonites and in sandstone sedimentary rocks.
The factors governing the formation of tv and cv layers in dioctahedral smectite are probably related to the layer composition and local order-disorder in the distribution of isomorphous octahedral cations, because there is no influence from fixed interlayer cations. In particular, the occurrence of Mg-OH-Mg cation arrangements is more favorable for the formation of cv montmorillonite layers.
Nickel Solubility and Precipitation in Soils: A Thermodynamic Study
- Edward Peltier, Ramakumar Allada, Alexandra Navrotsky, Donald L. Sparks
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 153-164
-
- Article
- Export citation
-
The formation of mixed-metal-Al layered double hydroxide (LDH) phases similar to hydrotalcite has been identified as a significant mechanism for immobilization of trace metals in some environmental systems. These precipitate phases become increasingly stable as they age, and their formation may therefore be an important pathway for sequestration of toxic metals in contaminated soils. However, the lack of thermodynamic data for LDH phases makes it difficult to model their behavior in natural systems. In this work, enthalpies of formation for Ni LDH phases with nitrate and sulfate interlayers were determined and compared to recently published data on carbonate interlayer LDHs. Differences in the identity of the anion interlayer resulted in substantial changes in the enthalpies of formation of the LDH phases, in the order of increasing enthalpy carbonate<sulfate<nitrate. Substitution of silica for carbonate resulted in an even more exothermic enthalpy of formation, confirming that silica substitution increases the stability of LDH precipitates. Both mechanical mixture and solid-solution models could be used to predict the thermodynamic properties of the LDH phases. Modeling results based on these thermodynamic data indicated that the formation of LDH phases on soil mineral substrates decreased Ni solubility compared to Ni(OH)2 over pH 5–9 when soluble Al is present in the soil substrate. Over time, both of these precipitate phases will transform to more stable Ni phyllosilicates.
Controlled Polymerization of Metanilic Anion within the Interlayer of NiAl Layered Double Hydroxide
- Xiaofei Tian, Min Wei, David G. Evans, Guoying Rao, Heli Yang
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 418-425
-
- Article
- Export citation
-
The controlled chemical oxidative polymerization of metanilic anion $(m{\rm{ - N}}{{\rm{H}}_2}{{\rm{C}}_6}{{\rm{H}}_4}{\rm{SO}}_3^ - )$
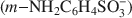 within the interlayer of NiAl layered double hydroxide was performed using, for the first time, ammonium persulfate as the oxidizing agent. The quantity of oxidizing agent required for control of the interlayer polymerization was investigated systematically and it was found that interleaved polyaniline sulfonic (PANIS) was present in different oxidation states and protonation levels when different quantities of external oxidizing agents were added. A mechanism for the oxidative polymerization of metanilic anion in NiAl layered double hydroxide is proposed, based on the intercalation of the oxidizing agent and the interlayer polymerization of monomer. The resulting PANIS/NiAl LDH composites were characterized by powder X-ray diffraction, ultraviolet-visible absorption spectra, Fourier transform infrared and X-ray photoelectron spectroscopy.
within the interlayer of NiAl layered double hydroxide was performed using, for the first time, ammonium persulfate as the oxidizing agent. The quantity of oxidizing agent required for control of the interlayer polymerization was investigated systematically and it was found that interleaved polyaniline sulfonic (PANIS) was present in different oxidation states and protonation levels when different quantities of external oxidizing agents were added. A mechanism for the oxidative polymerization of metanilic anion in NiAl layered double hydroxide is proposed, based on the intercalation of the oxidizing agent and the interlayer polymerization of monomer. The resulting PANIS/NiAl LDH composites were characterized by powder X-ray diffraction, ultraviolet-visible absorption spectra, Fourier transform infrared and X-ray photoelectron spectroscopy.
Mineralogy and Geochemistry of the Host-Rock Alterations Associated with the Shea Creek Unconformity-Type Uranium Deposits (Athabasca Basin, Saskatchewan, Canada). Part 2. Regional-Scale Spatial Distribution of the Athabasca Group Sandstone Matrix Minerals
- Philippe Kister, Emmanuel Laverret, David Quirt, Michel Cuney, Patricia Patrier Mas, Daniel Beaufort, Patrice Bruneton
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 295-313
-
- Article
- Export citation
-
The spatial distribution of the dominant matrix minerals present in the middle-Proterozoic Athabasca Group sandstone (kaolin, illite, sudoite, dravite, hematite) was studied at a regional scale in the Shea Creek region (Saskatchewan, Canada), in which two epigenetic unconformity-type uranium deposits have been discovered. 3D models of matrix mineral distribution were derived from normative mineral calculations and 3D interpolation using whole-rock geochemical analyses of sandstone samples collected from both mineralized and barren areas. The calculations were constrained by information obtained from petrographic and crystal-chemical clay mineralogical studies on representative samples. The 3D mineral distribution models were compared to the lithostratigraphy and structural features of the Athabasca Group sandstone to ascertain the source and mobility of the main elements involved in the sandstone host-rock alteration processes related to the U mineralization. The distribution of Al is conformable with the lithostratigraphy throughout the studied area, regardless of proximity to basement-rooted structures and U ore bodies. The distribution of illite displays similar features, but the intensity of the illitization of kaolin decreases with increasing distance from the structures and U ore bodies. Hematite bleaching and neoformation of sudoite and dravite were restricted to the vicinity of the fault zones above the U ore bodies. The spatial configurations of the mineral anomalies show that syn-ore fluids flowed from the basement towards the sandstone cover via the fault zones, as described in current metallogenic models. Although Al remained immobile (mass transfer), the anomalous K, B and Mg present in the host-rock alteration haloes were probably imported from the basement rocks (mass transport). Unlike B and Mg, K migrated laterally at least several kilometers from the basement-rooted faults. The mineral distribution models were used to quantify the volume of altered sandstone (10−2−10−1 km3) and the amounts of K, Mg and B which were imported to the alteration haloes above the Shea Creek U ore bodies: 186,000 t of K, 66,000 t of Mg, and 11,000 t of B above the Anne ore body, and 24,000 t of K, 185,000 t of Mg, and a similar 11,000 t of B above the Colette ore body.
Properties of surface-modified colloidal particles
- Joan M. Breiner, Michael A. Anderson, Harry W. K. Tom, Robert C. Graham
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 12-24
-
- Article
- Export citation
-
The reactivity of colloidal particles is regulated by their surface properties. These properties affect the wettability, flocculation-dispersion characteristics, ion exchange, sorption capacities and transport of inorganic colloids. Most studies have focused on hydrophilic, charged-particle surfaces, often ignoring the alterations in surface properties produced by the adsorption of natural organic matter, surfactants and other compounds. Adsorption of these substances can potentially render a surface substantially more hydrophobic. Nevertheless, comparatively little is known about changes in surface properties and reactivity of minerals upon sorption of hydrophobic organic compounds. In this study, the properties of four minerals (kaolinite, pyrophyllite, montmorillonite and Min-U-Sil®) and two inorganic materials (X-ray amorphous Al hydroxide and X-ray amorphous Si oxide) were compared before and after treatment with the common silylating agent, trimethylchlorosilane (TMCS). The samples were characterized by measurements of total carbon, cation exchange capacity (CEC), particle size, specific surface area (SSA), electrophoretic mobility, contact angle, particle aggregation, and by X-ray diffraction and diffuse reflectance infrared spectroscopy. For the layer silicates, surface coverage was limited to ∼2% trimethyl silane (TMSi). TMSi covered 7.5% of the Min-U-Sil® surface and 33% of the X-ray amorphous Si oxide. Treatment did not affect the structure of the minerals but reduced the CEC, SSA and electrophoretic mobilities. Water contact angles increased to between 18 and 114° with treatment. While the apolar characteristic of the surfaces decreased minimally with treatment, the Lewis acid/base properties were substantially reduced and interfacial free energy shifted from positive to negative values indicating a more hydrophobic surface character. For all the samples except kaolinite, these changes affected the stability of the colloids in suspension depending upon solution pH. Although the grafting of TMSi altered colloidal mineral surface properties and increased their hydrophobicity, these changes were not sufficient to predict colloid aggregation behavior.
Hydrothermal Synthesis of Mg-Rich and Mg-Ni-Rich Kaolinite
- Maria Bentabol, Maria Dolores Ruiz Cruz, Francisco Javier Huertas, Jose Linares
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 667-677
-
- Article
- Export citation
-
Mg-rich kaolinite and Mg+Ni-rich kaolinite have been synthesized in hydrothermal experiments (200 and 400°C) from poorly crystalline kaolinite and Mg- and Mg+Ni-bearing solutions. Al-rich serpentine and Al-rich chlorite were obtained as sub-products of the reactions. The formation of these phases occurred through a dissolution-precipitation mechanism that led to spherical kaolinite after short reaction times. A morphological evolution towards platy particles and stacks occurred at increasing run times.
Identification of the several phases was carried out using a combination of X-ray diffraction and transmission/analytical electron microscopy. Analytical data indicate that the Mg content in kaolinite increased as a function of the reaction time and temperature, reaching up to 0.46 atoms per half formula unit (a.p.h.f.u.). The measured (Mg+Ni) content reached up to 0.56 a.p.h.f.u.. Both the gradual increase of the b-cell parameter of kaolinite at increasing Mg contents and the presence of new bands on the FTIR spectra of the synthesized kaolinite point to a Mg-for-Al replacement in the octahedral sheet rather than to the presence of serpentine-like layers interstratified in the kaolinite structure.
Clay Minerals Formed During Propylitic Alteration of a Granite and Their Influence on Primary Porosity: A Multi-Scale Approach
- M. Cassiaux, D. Proust, M. Siitari-Kauppi, P. Sardini, Y. Leutsch
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 541-554
-
- Article
- Export citation
-
The porosity of a propylitized granite from Charroux (France), with no fractures or sealed fractures, increases by more than four times from the unaltered (0.3%) to the altered rock (1.4%). This evolution results from several local porosity modifications which occur at different scales in the rock: (1) at the core scale, from 10−1 to 10−5 m, where rock porosity changes as a function of rock-forming mineralogical composition; (2) at the mineral scale, from 10−3 to 10−7 m, where porosity depends both on the nature of the rock-forming mineral and its clay mineral alteration. Mineralogical and porosity data collected from the granite using a mineralogical map (after chemical staining and scanning electron microscopy images combined with autoradiographs) indicate that (1) the ferromagnesian rock-forming minerals — biotite and magnesiohornblende — act as the main porosity source in the unaltered granite, and (2) the nature of the clay minerals replacing rock-forming minerals in the altered granite appears to control the porosity value through two major alteration processes: chloritization and phengitization which affect the ferromagnesian minerals and produce non-porous chloritic and porous phengitic areas, respectively, at the studied scales. The observation that incipient porosity formation in granites is strongly linked to the pathway of ferromagnesian silicate alteration and subsequent clay mineral formation underlines the need to study parent-rock texture and mineralogy and their effects on subsequent near-surface weathering of granites.
Comparison of K-Ar Ages of Diagenetic Illite-Smectite to the Age of a Chemical Remanent Magnetization (CRM): An Example from the Isle of Skye, Scotland
- W. Crawford Elliott, Ankan Basu, J. Marion Wampler, R. Douglas Elmore, Georg H. Grathoff
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 314-323
-
- Article
- Export citation
-
The clay fractions of Jurassic marls in the Great Estuarine Group in southern Isle of Skye are composed of mixed-layered illite-smectite (I-S) with large percentages (>85%) of illite layers, kaolinite, and generally smaller amounts of chlorite. These marls have not been buried to the depths normally required to convert smectite to illite-rich I-S, so it is possible that the conversion was in response to heat and hydrothermal fluids from nearby early Tertiary igneous activity ∼55 Ma ago. The large percentages of illite layers in I-S, the Środoń intensity ratios, and the Kübler index values appear to be consistent with the formation of diagenetic I-S as a result of relatively brief heating caused by igneous activity. The Jurassic rocks in southern Skye contain a secondary chemical remanent magnetization (CRM) that resides in magnetite and formed at approximately the same time as the Tertiary igneous rocks on Skye. K-Ar age values for I-S based on illite age analysis have been determined to test the hypothesis that the CRM was acquired coincidently with the smectite-to-illite conversion. However, linear extrapolation of K-Ar age vs. percentage of 2M1 polytype (detrital illite) from one marl (EL-6) yields an estimate for the age of diagenetic illite of 106 Ma, which is close to the measured age of the finest subfraction (108 Ma). These estimated and measured age values, however, could be substantially greater than the true age of the diagenetic illite in I-S because of the presence of detrital 1Md illite that was recycled from early Paleozoic shales and whose abundance relative to the diagenetic I-S may have been enhanced because the diagenetic fluid had a low K/Na ratio, limiting the amount of diagenetic illite formed. Nevertheless, most of the illite in the Elgol marls (80% or more in the finest fractions) must be diagenetic and probably formed in response to the early Tertiary magmatism.
Adhered Zeolite Preparation on and Within a Muscovite Mica by Hydrothermal Growth
- Christopher D. Johnson, Anthony J. Mallon, Fred Worrall
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 678-688
-
- Article
- Export citation
-
Zeolites and other open framework materials provide a powerful tool for remediation and solidification of a range of cationic wastes (e.g.${\rm{NH}}_4^ + $
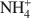 , Pb2+) due to the combined properties of large surface area and cation exchange capacity. However, practical barriers exist to the continued expansion of their use, including handling issues related to the fine particle size, and continued ion exchange following waste adsorption. This study examines the synthesis and characterization of zeolites adhered to a muscovite mica wafer, in order to assess if practical benefit can be derived from the preparation of layered composite materials. The paper demonstrates that increased metal adsorption, as demonstrated by surface chemical composition, can be induced in regions by growth of zeolite on and within the lamellar structure of the matrix. X-ray diffraction studies suggest that a site-specific crystallization mechanism controls the zeolite type and extent of growth, thereby reducing control over the zeolites prepared. However, although increased adsorption has been introduced to the mica, the amount of zeolite added is small (<50 mg per gram of muscovite), and thus any adsorption is very limited.
, Pb2+) due to the combined properties of large surface area and cation exchange capacity. However, practical barriers exist to the continued expansion of their use, including handling issues related to the fine particle size, and continued ion exchange following waste adsorption. This study examines the synthesis and characterization of zeolites adhered to a muscovite mica wafer, in order to assess if practical benefit can be derived from the preparation of layered composite materials. The paper demonstrates that increased metal adsorption, as demonstrated by surface chemical composition, can be induced in regions by growth of zeolite on and within the lamellar structure of the matrix. X-ray diffraction studies suggest that a site-specific crystallization mechanism controls the zeolite type and extent of growth, thereby reducing control over the zeolites prepared. However, although increased adsorption has been introduced to the mica, the amount of zeolite added is small (<50 mg per gram of muscovite), and thus any adsorption is very limited.
Sorption of Nitroaromatics by Ammonium- and Organic Ammonium-Exchanged Smectite: Shifts from Adsorption/Complexation to a Partition-Dominated Process
- Michael G. Roberts, Hui Li, Brian J. Teppen, Stephen A. Boyd
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 426-434
-
- Article
- Export citation
-
Nitroaromatic compounds (NACs) are components of munitions commonly found as soil contaminants at military training sites and elsewhere. These compounds pose possible threats to human health and ecological systems. Recent studies indicate that these compounds are strongly retained by smectite clays. The adsorption mechanisms are not fully reconciled, but it is known that the type of exchangeable cation strongly affects NAC affinity for smectites. This study examined the sorption of 1,3-dinitrobenzene, 2,4-dinitrotoluene and naphthalene from water by a smectite clay (SWy-2) saturated with ammonium, tetramethylammonium (TMA), trimethylphenylammonium (TMPA) and hexadecyltrimethylammonium (HDTMA). In all cases, we observed greater sorption of 2,4-dinitrotoluene compared with 1,3-dinitrobenzene. The sorption isotherms for 2,4-dinitrotoluene and 1,3-dinitrobenzene displayed a concave-downward curve for NH4-SWy-2 and TMA-SWy-2, whereas the isotherms for sorption of HDTMA-SWy-2 and TMPA-SWy-2 were essentially linear. The magnitude of sorption followed the order: NH4-SWy-2 > TMA-SWy-2 > TMPA-SWy-2 > HDTMA-SWy-2 for both compounds. The greater affinity of NACs for NH4- and TMA-SWy-2 is due in part to complex formation between the exchangeable cation and −NO2 groups. These clays also provide near optimal interlayer distances that approximate the molecular thickness of NACs hence promoting the simultaneous interaction of the planar aromatic rings with opposing siloxane surfaces and solute dehydration. Both processes are energetically favorable. In HDTMA-SWy-2, sorption of all solutes is via a partition-dominated process. Solute competition (diminished uptake of one solute in the presence of a second) was observed for TMA-SWy-2 but not HDTMA-SWy-2. This is consistent with an adsorptive mechanism for TMA-SWy-2 and a partitioning mechanism for HDTMA-SWy-2. This study demonstrates that the dominant molecular mechanism of NAC sorption by smectite changes fundamentally from complexation between −NO2 groups and the exchangeable cation (viz. NH4 and TMA) to partitioning for a systematic series of ammonium and quaternary ammonium cations in which the locus of positive charge (the central N atom) is progressively shielded by organic moieties of increasing size.
Geology, Mineralogy and Origin of Clay Minerals of the Pliocene Fluvial-Lacustrine Deposits in the Cappadocian Volcanic Province, Central Anatolia, Turkey
- Ali Gürel, Selahattın Kadır
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 555-570
-
- Article
- Export citation
-
The Güzelöz-İncesu Plateaus are situated in the central and eastern parts of the Cappadocian volcanic province (central Anatolia). This province contains many ignimbrite levels, andesite, basalt intercalated with several paleosols, calcrete, carbonate, fluvial sediments, diatomaceous clayey sediments and pyroclastic sedimentary levels. The presence of mottling, sesquioxide, root traces, rhizoids and burrows in continuous, finely bedded and laminated sediments, paleosols, calcrete, the occurrence of bone- and teeth-bearing reworked pyroclastic materials, and the description of the lithofacies in the study area indicate fluvial and shallow-lake environments. These environments are dominated by smectite and illite, with traces of kaolinite, associated mainly with plagioclase, K-feldspar, quartz, calcite, opal-CT, pyroxene (diopside, rare hypersthene), and locally trace amounts of gypsum and sepiolite. Smectite predominates in paleosols and calcrete units, and generally increases upwards in the profiles, coinciding with a gradual increase in the degree of alteration. Partial to complete alteration of plagioclase, K-feldspar, pyroxene and partial devitrification of glass-shard particles in pyroclastic rocks, development of microsparitic to sparitic cement comprising euhedral rhombic calcite crystals between irregular clay nodules in paleosol and calcrete samples, along with the occurrence of dogtooth-type sparitic crystals in fractures, desiccation cracks and geopetal-type fenestrae, indicate alternating periods of drought and wet, resulting in the development of paleosols and calcretes. Micromorphological development of spongiform smectite on mainly relict feldspar and, locally, on glass shards, indicates an authigenic origin, whereas illite formed either authigenically or by conversion of smectite to illite-smectite.
Use of atomic force microscopy for examining wet clay
- Arunkumar Selvam, Chun Hwa See, Brian Barkdoll, Shyam Prasad, John O’Haver
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 25-28
-
- Article
- Export citation
-
Clays and their composites have been widely used for secondary containment walls for underground storage tanks and landfills. The pore-size changes occurring in the clay have a profound effect on its permeability. This study presents a new method for evaluating the use of an atomic force microscope (AFM) for studying wet clay in a non-aqueous state in order to determine the pore-size of clay at various water contents, a type of study typically performed by the more expensive environmental scanning electronic microscope. The method consists of mounting a sponge saturated with water under the sample in order to prevent drying by the heat generated by the AFM electronics. The micro-scale AFM image results show that the clay-particle separations reduce linearly as the water content increases. This change in pore-size is postulated to be attributed to the reduction in the size of the diffuse double layer and more extensive hydrogen bonds between clay particles and bipolar water molecules. The AFM was not able to produce nano-scale images due to excessive adhesion between the cantilever arm and the wet clay sample.
Heating Fe Oxide-Rich Soils Increases the Dissolution Rate of Metals
- Nicolas Perrier, Robert J. Gilkes, Fabrice Colin
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 165-175
-
- Article
- Export citation
-
Evidence for fire affecting the solubility of metals in Fe oxide-rich Oxisols of the Koniambo Massif of New Caledonia is presented. Acid-dissolution studies showed that Ni, Al and Cr are substituted for Fe in the structure of the Fe oxides. Thermal dehydroxylation of goethite under oxidizing conditions led to the formation of hematite and to the migration of some of these metals towards the surface of hematite crystals as indicated by their enhanced release during the early stage of dissolution. Dehydroxylation of goethite under reducing conditions led to the formation of hematite and maghemite. Nickel and Al were released preferentially during the early stages of dissolution whereas Cr was not released preferentially and may be uniformly incorporated within maghemite and hematite crystals. These results have significance to the mineral-processing industry, to geochemical exploration and to the availability of these metals to plants growing on burnt soils.
The Smectite-to-Disordered Kaolinite Transition in a Tropical Soil Chronosequence, Pacific Coast, Costa Rica
- G. Burch Fisher, Peter C. Ryan
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 571-586
-
- Article
- Export citation
-
Soils developed on Quaternary fluvial fill terraces in the humid tropics of Costa Rica display progressive changes in mineral assemblage, chemical composition and particle size with age. Clay minerals from B horizons of active floodplains are predominantly smectite with lesser amounts of disordered kaolinite. B horizons in 5 to 10 ka soils consist of sub-equal amounts of smectite and disordered kaolinite, and soils on 37–125 ka terraces consist of disordered kaolinite with only traces of smectite. The composition of the smectite, as determined by EDX scans of smectite-rich pore space, is [(Mg0.2,Ca0.1)(Fe0.6Al1.4)(Si3.6Al0.4)O10(OH)2], consistent with ferruginous beidellite.
Bulk mineral assemblage varies from a smectite-plagioclase-augite-quartz-magnetite assemblage in ⩽ 10 ka terrace soils to a disordered kaolinite-goethite-hematite-quartz-magnetite assemblage in ⩾37 ka terrace soils. Leaching results in rapid loss of soluble base cations and residual concentration of Ti and Zr indicates mass losses of ∼50% by chemical denudation by 125 ka. Plots of terrace age vs. various measures of clay mineralogy, chemical composition, and particle size produce parabolic curves consistent with rapid chemical weathering pre-37 ka and slower to imperceptible rates of change from 37 to 125 ka. For some pedogenic properties, particularly particle size and concentrations of base cations and Zr, soils appear to reach steady-state conditions within 37 ka.
These results were applied to interpretation of landscape evolution in this tectonically active region by: (1) facilitating identification of two Holocene (5 ka and 10 ka) terraces on the Esterillos Block 5–30 m above sea level (masl), and two Pleistocene terraces ⩾ 125 ka on the Parrita Block 30 masl, and, in turn, (2) documenting uplift rates as high as 4.4 m/ka between 37 and 10 ka on the Esterillos Block, and as low as 0.1 m/ka over the past 125 ka on the adjacent Parrita Block. These findings are consistent with previous work indicating that the subduction of anomalous bathymetric features at the Middle America Trench is having a significant impact on fore-arc dynamics and topography over relatively short geological time periods and spatial scales.
Thermal and Mineral Properties of Al-, Cr-, Mn-, Ni- and Ti-Substituted Goethite
- M. A. Wells, R. W. Fitzpatrick, R. J. Gilkes
-
- Published online by Cambridge University Press:
- 01 January 2024, pp. 176-194
-
- Article
- Export citation
-
Mineralogical and thermal characteristics of synthetic Al-, Cr-, Mn-, Ni- and Ti-bearing goethites, synthesized via alkaline hydrolysis of metal-ferrihydrite gels, were investigated by powder X-ray diffraction and differential thermal analysis. Shifts in unit-cell dimensions were consistent with size of substituent metal ions and confirmed the incorporation of Al3+, Cr3+, Mn3+, Ni2+ and Ti4+ in the goethite structure. A weight loss of 6.2 wt.% for goethite containing 12.2 mol.% Ti, being significantly less than for stoichiometric goethite, is consistent with the replacement of Fe by Ti in the goethite structure coupled with the substitution of O2− ions for OH− (i.e. proton loss). These data provide the first confirmation of the direct replacement of Fe by Ti within goethite. Formation of multiple dehydroxylation endotherms for goethite containing 4.5 mol.% Al, 15.3 mol.% Mn and 12.2 mol.% Ti was not attributed to the decomposition of surface OH groups or related simply to the crystallinity of precursor goethite (‘high-a’ vs. ‘low-a’) as defined by the magnitude of a. Instead, endotherm doublet formation was associated with weight loss due to the dehydroxylation of goethite remaining after initial phase transformation to protohematite and to the evolution of OH− associated with the rapid increase in crystallite size of protohematite directed primarily along the a direction. Development of the first endotherm is due to initial dehydroxylation and transformation to protohematite. With continued heating of well ordered goethite or goethite containing moderate to high levels of substituent cations, domain growth along the a direction is delayed or inhibited to a critical point that provides enough thermal energy to enable goethite transformation to proceed to completion and for proto-hematite domain growth to occur. This results in the formation of a second endotherm. For less well ordered goethite and/or goethite containing only low levels of foreign metal cations, protohematite domain growth is not inhibited and proceeds continuously with heating to give only a single endotherm.