Poster Presentation - Poster Presentation
Antibiotic Stewardship
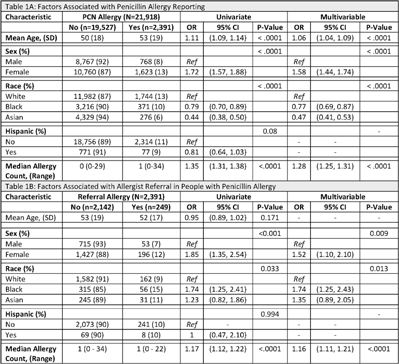
Racial and ethnic differences in penicillin allergy reporting and allergist referral
- Charles Bornmann, Christina Ortiz, Rubeen Guardado, Joseph Gillis, Jr, Kristin Huang, Kimberly Blumenthal, Shira Doron, Maureen Campion, Alysse Wurcel
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s23
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antimicrobial resistance (AMR) is a global public health crisis. A key strategy to combat AMR is to use targeted antibiotics, which is difficult in patients who report an allergy to penicillin. Increased risk for resistant infections, mortality, and healthcare costs are associated with penicillin allergies; however, up to 90% of those with a reported penicillin allergy do not have a true allergy. We investigated racial and ethnic differences related to penicillin allergy delabeling by analyzing rates of penicillin allergy reporting and referral for allergist consultation. Methods: Tufts Medical Center is a teaching medical center in Boston, Massachusetts. This study cohort contains all patients seen in 2019 by clinicians at Primary Care Boston, the main primary care practice at Tufts Medical Center. Demographic data, documented allergies, and referral history were collected from the electronic medical record. We performed univariate and multivariable analyses using logistic regression models. Covariates found to be statistically significant (P < .05) in the univariate analyses were included in the multivariable model. Results:In total, 2,391 (11%) patients reported a penicillin allergy, but only 249 (10%) were referred to an allergist (Table 1). Black patients and Asian patients were less likely to report a penicillin allergy than White patients. We detected no differences related to Hispanic ethnicity. Black patients with penicillin allergy were more likely to be referred to an allergist. Conclusions: There were low rates of allergist referral for penicillin allergy delabeling in this cohort. We identified racial differences in both penicillin allergy reporting and allergist referral. Allergist consultation is an important opportunity to combat AMR and should be considered for all patients reporting a penicillin allergy. Future work should evaluate equitable access to allergy delabeling resources.
Funding: None
Disclosures: None
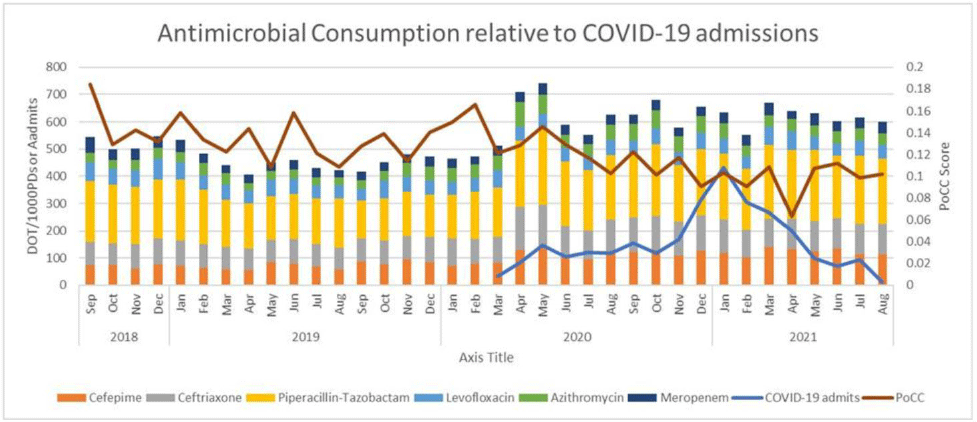
Antimicrobial use patterns during the COVID-19 pandemic at an academic medical center
- Jacob Pierce, Erin Deja, Kimberly Lee, Michelle Doll, Michael Stevens
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s23-s24
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The COVID-19 pandemic has made a significant impact on antimicrobial use patterns across health systems. We have described antibiotic use patterns at an academic medical center in Richmond, Virginia, before and after the onset of COVID-19. We also examined the impact on the proportional consumption of carbapenems (PoCC) metric. PoCC represents meropenem utilization relative to the narrower-spectrum antipseudomonal agents cefepime and piperacillin-tazobactam. Our institution practices antimicrobial restriction for meropenem. All other antibiotics included in the study data can be freely ordered by any provider. Methods: We evaluated antimicrobial use data from September 2018 through August 2021 using days of therapy (DOT) per 1,000 patient days. We included 18 months of data before and after the first recorded COVID-19 admission at our institution in March 2020. Mean comparisons were performed using the Welch 2-sample t test. The Bonferonni correction for multiple comparisons was utilized to determine significance with an initial baseline α of 0.05. All data analyses were performed using R software (R Foundation for Statistical Computing, Vienna, Austria, 2021). Results: Normality was evaluated with QQ-plots and all data demonstrated normality. Bonferroni correction produced an adjusted α value of 0.007. We detected significant increases in the use of cefepime, piperacillin-tazobactam, ceftriaxone, and azithromycin following the onset of the COVID-19 pandemic. We noted a significant decrease in the PoCC metric during this period. No significant change was noted for levofloxacin or meropenem. Conclusions: The COVID-19 pandemic produced significant changes in antimicrobial use patterns at our institution. We noted statistically significant increases in bacterial community-acquired pneumonia-focused antibiotics (ceftriaxone and azithromycin). We observed significant increases for cefepime and piperacillin-tazobactam. Interestingly, relative utilization of carbapenems as measured by the PoCC metric continued to decrease during this time. This trend was primarily driven by increases in cefepime and piperacillin-tazobactam utilization while meropenem utilization remained relatively constant. This study highlights the importance of looking at normalized antibiotic consumption data and not relative-use data alone.
Funding: None
Disclosures: None
Recommendations for antimicrobial stewardship during end-of-life patient care
- Erin Balkenende, Cassie Goedken, Daniel Livorsi, Karleen Giannitrapani, Matthew McCaa, Eli Perencevich
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s24
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antimicrobials are frequently used during end-of-life care and may be prescribed without a clear clinical indication. Overuse of antimicrobials is a major public health concern because of the development of multidrug resistant organisms (MDROs). Antimicrobial stewardship programs are associated with reductions in antibiotic resistance and antibiotic-associated adverse events. We sought to identify and describe opportunities to successfully incorporate stewardship strategies into end-of-life care. Methods: We completed semistructured interviews with 15 healthcare providers at 2 VA medical centers, 1 inpatient setting and 1 long-term care setting. Interviews were conducted via telephone between November 2020 and June 2021 and covered topics related to antibiotic prescribing for hospice and palliative-care patients, including how to improve antimicrobial stewardship during the end-of-life period. We targeted healthcare providers who are involved in prescribing antibiotics during the end-of-life period, including hospitalists, infectious disease physicians, palliative care and hospice physicians, and pharmacists. All interviews were recorded, transcribed, and analyzed using consensus-based inductive and deductive coding. Results: End-of-life care, particularly hospice care, was described as an underutilized resource for patients, who are often enrolled in their final days of life rather than earlier in the dying process. Even at facilities with established antimicrobial stewardship programs, healthcare providers interviewed believed that opportunities for antimicrobial stewardship in the hospice and palliative care settings were missed. Recommendations for how stewardship should be incorporated in end-of-life care included receiving feedback on antimicrobial prescribing, increasing pharmacist involvement in prescribing decisions, and targeted education for providers on end-of-life care, including the value of shared decision making with patients around antibiotic use. Conclusions: Improved antibiotic prescribing during end-of-life care is critical in the effort to combat antimicrobial resistance. Healthcare providers discussed antimicrobial stewardship activities during end-of-life patient care as a potential avenue to improve appropriate antibiotic prescribing. Future research should evaluate the feasibility and effectiveness of incorporating these strategies into end-of-life patient care.
Funding: None
Disclosures: None
Assessment of antibiotic appropriateness in hospitalized veterans with COVID-19 in the VA MidSouth Healthcare Network (VISN9)
- Derek Forster, Morgan Johnson, Milner Staub, Jessica Bennett, Hans Scheerenberger, Angela Kaucher, Neena Thomas-Gosain, Kelly Davis
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s24-s25
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Bacterial coinfections with COVID-19 appear to be rare, yet antibiotic use in this population is high. Limited guidance is available regarding the use of antibiotics in these patients. In response, a multidisciplinary group of physicians and pharmacists from 5 VISN9 facilities developed a guideline for the use of antibiotics with COVID-19 in July 2021. This guideline created a network-wide standard for antibiotic use and facilitates the assessment of antibiotic appropriateness in hospitalized veterans with COVID-19. Methods: In this observational, cross-sectional study, we reviewed veterans diagnosed with COVID-19 from August 1 through September 30, 2021, who were admitted to VISN9 facilities. Use of antibiotics was assessed during the first 4 days of admission. If antibiotics were prescribed, their use was determined to be appropriate or inappropriate based on the presence or absence of a finding concerning for bacterial coinfection as outlined in the guideline (Table 1). Additional data including procalcitonin results as well as positive sputum cultures were collected. Results: In total, 377 veterans were admitted for COVID-19 during the study period. Among them, 42 veterans (11%) received antibiotics for nonrespiratory infections and were removed from this analysis. Of the remaining 335 veterans, 229 (68%) received antibiotics and 116 (51%) of those met guideline criteria that were concerning for bacterial coinfection. Additionally, 32 (14%) of the 229 veterans who received antibiotics had >1 finding concerning for bacterial coinfection. Procalcitonin levels were obtained in 97 (42%) of 229. Only 33 veterans (14%) who received antibiotics had an elevated procalcitonin, and only 19 (8%) had a positive sputum culture. Conclusions: Antibiotic use was common in hospitalized veterans with COVID-19 in VISN9 facilities. This results are comparable to findings in the published literature. Among those receiving antibiotics early in their hospitalization, half were considered appropriate based on our guideline. Quality improvement initiatives are needed to improve implementation of the network guideline to reduce the overuse of antibiotics for management of COVID-19. Additionally, procalcitonin may be a helpful tool for hospitalized veterans with COVID-19.
Funding: None
Disclosures: None

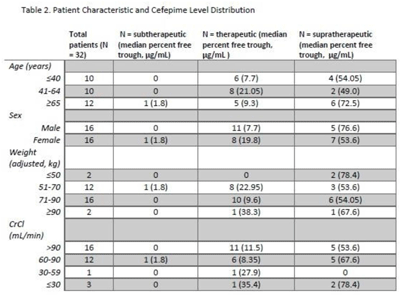
Assessing the association between cefepime percentage free trough level and neurotoxicity
- Aleena Zia, Armisha Desai, Molly Tieman, Haley Pritchard
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s25
-
- Article
-
- You have access Access
- Open access
- Export citation
-
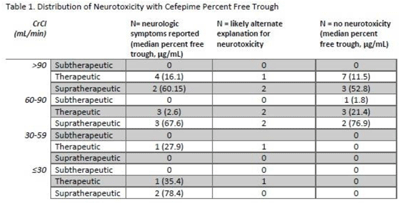
Background: Cefepime has a known association with neurotoxicity due to its ability to cross the blood–brain barrier. The symptoms of neurotoxicity are highly variable. It has been postulated that cefepime neurotoxicity is associated with elevated levels of the drug. However, studies assessing for an association between serum drug level and the incidence of neurotoxicity have yet to establish a consistent threshold. We assessed serum cefepime levels and incidence of neurotoxicity to help develop a dosing strategy to minimize adverse effects. Method: In total, 32 inpatients admitted from January 2019 to November 2021 who received cefepime according to institutional standard dosing regimens for at least 72 hours were reviewed by infectious diseases pharmacists who obtained serum cefepime levels and performed pharmacokinetic analyses to obtain percentage free trough levels. Cefepime percentage free trough levels were defined as therapeutic if they were above the known minimum inhibitory concentration (MIC) of the treated organism and were <40 μg/mL. Patient charts were reviewed for clinical findings consistent with cefepime-induced neurotoxicity. Numerical and statistical analyses were performed to assess factors with a significant association with neurotoxicity. Results: Overall, 16 (47.1%) patients showed some evidence of neurotoxicity, 9 (56.3%) of whom had a likely alternate clinical cause of symptoms (Table 1). We did observe that patients with creatinine clearance <60 mL/min were more likely to have symptoms concerning for neurotoxicity. Conclusions: Cefepime percentage free trough levels were highly variable, and no association with neurotoxicity was observed. Patients with decreased creatinine clearance were significantly more likely to develop neurologic findings consistent with cefepime-induced neurotoxicity. Further study is needed to establish a relationship between cefepime pharmacokinetic values and incidence of neurotoxicity.
Funding: None
Disclosures: None
Implementing Leading antimicrobial stewardship practices in United States hospitals – A qualitative study
- Salome Chitavi, Mike Kohut, Barbara Braun, Meghann Adams, David Hyun
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s25-s26
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: In May 2018, The Joint Commission, The Pew Charitable Trusts, and the CDC cosponsored a meeting of experts who identified 6 evidence-based leading practices that antimicrobial stewardship programs (ASPs) should be doing beyond having basic infrastructure for improving antibiotic prescribing. The Joint Commission Department of Research working with external experts in 2020 conducted a prevalence study to assess what proportion of Joint Commission-accredited hospitals had implemented the 6 leading practices identified (results presented at SHEA Spring 2021). In this qualitative study, we collected information about how hospitals implemented ASP leading practices to identify facilitators and barriers to implementation among diverse hospitals. Methods: We conducted in-depth telephone interviews with a subset of ASP leaders from hospitals that participated in the 2020 prevalence study. We used purposive sampling to select 30 hospitals from 288 hospitals based on leading practices implemented, hospital size, and system membership. An experienced qualitative researcher (M.K.) not previously affiliated with the Joint Commission interviewed all participants using a semistructured interview guide. The framework method of analysis was used to review and organize data. We used the constant comparative approach to ensure that factors were not missed. Each transcript was reviewed by at least 2 researchers who compared coded findings in group discussion sessions. Two researchers independently identified key factors and combined findings following discussion and review. We focused on super factors that are relevant to implementing multiple leading practices. Results: ASP leaders from 30 hospitals were interviewed. Participating hospitals were evenly distributed across hospital size (10 small, 10 medium, 10 large) and membership in a health system (16 system, 14 nonsystem). At least 14, (46.7%) interviewees had pharmacist in their title; 11 (36.7%) had pharmacist-antimicrobial stewardship; and 5 (16.6%) had other titles (eg, infection preventionist). Super factors included ASP team capacity, ID expertise, having a physician champion, relationships with clinicians and relevant departments, structure of electronic health records, adequate software, and information technology resources. Small and rural nonsystem hospitals often lacked resources related to ID expertise, dedicated staff, and software tools, whereas hospitals that belong to a system benefit from centralized ID expertise and technical infrastructure provided. Conclusions: Specific factors related to personnel, relationships and IT resources have an outsized impact on implementing multiple leading antimicrobial stewardship practices in hospitals. Hospital ASPs could benefit by targeting resources toward these areas.
Funding: None
Disclosures: None
Systematic review of antibiotic stewardship interventions for urinary tract infection management in the ambulatory setting
- Sonal Munsiff, Alexandra Sakai, Ghinwa Dumyati
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s26
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Urinary tract infections (UTIs) are common indications for antibiotics in ambulatory setting, and inappropriate use is prevalent. Fluoroquinolones account for 40% of antibiotics prescribed for uncomplicated UTIs, despite clinical guidance against their use as firstline agents. We conducted a systematic review to determine which antibiotic stewardship intervention(s) are effective in improving antibiotic prescribing for UTIs in the ambulatory setting. Methods: Following PRISMA guidelines, English-language literature from 1995 to September 21, 2021, was searched for articles about antimicrobial stewardship, UTI, and ambulatory setting from PubMed, Embase, and Central. Additional articles were identified from authors’ collections and references of pertinent articles. Studies were included if the authors implemented intervention targeting adults 18 years and older in outpatient setting (excluding emergency departments). Interventions were categorized into Guideline Development and Dissemination (GDD), Audit and Feedback, Clinical Decision Support System (CDSS), and Multimodal Interventions. Results: The literature search identified 1,899 papers; 14 papers were included in this review; and 4 additional papers were identified from other sources. The main interventions were GDD in 6 studies, audit and feedback in 3 studies, CDSS in 4 studies, and multimodal interventions in 5 stidues. These studies had heterogeneity of the practice settings and interventions. Moreover, 11 studies targeted primary care, 2 studies targeted urgent care, 1 study targeted both primary and urgent care, 2 studies were conducted in spinal cord injury clinics, and 2 studies were conducted in hospital-wide outpatient sites. Outcomes included (1) statistically significant increase in guideline-concordant antibiotic prescribing in 12 studies (range, 4.6%–246%); (2) statistically significant decrease in fluoroquinolone prescriptions (range, 9.1%–86.3%) in 7 of 9 studies focusing on fluoroquinolones; (3) significant decreases in drug resistance in urine pathogens in 2 studies that evaluated this. Provider education, in conjunction with passive CDSS tools, such as integrating order sets for UTI prescriptions with prefilled instructions into electronic medical records appeared most beneficial. Several studies have investigated negative impact and have found no increase in retreatment rates or worse outcomes. Conclusions: Our systematic literature review identified a limited number of studies with a variety of interventions that improved antibiotic use for UTIs in the ambulatory care setting. Provider education, in conjunction with CDSS tools, can be less time-consuming than audit and feedback and can target a large number of providers and practices. Future studies need to address sustainability over longer periods and should target specialty clinic populations because they have high burden of patients with multidrug-resistant UTI organisms.
Funding: None
Disclosures: None
Antibiotic use and impact on illness course in children with influenza-like-illness in the emergency department
- Nicole Poole, Sean O’Leary, Suchitra Rao, Krithika Suresh, Angela Moss
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s26
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Child absenteeism from childcare or school leads to economic loss from parental work absenteeism, overutilization of acute-care resources, and excess medicalization of children with minor illnesses. We sought to determine the difference in days missed from childcare or school and days of illness for children with influenza-like illness (ILI) in the emergency department (ED) who are or are not prescribed an antibiotic. Methods: We conducted a secondary data analysis of a prospective randomized control trial evaluating the impact of rapid molecular testing on provider decision making. The study included children aged 2 months–12 years attending childcare or school seen in the ED from December 2018 through December 2019 with ILI (CDC definition) with parental survey completion 10 days after their ED visit. The primary exposure was receipt of antibiotics over the course of illness, which was assessed by chart review and parent survey. The primary outcome was number of days missed from class. The secondary outcome was number of days of illness after initial ED visit. Wilcoxon tests were used to compare missed class days or illness days by antibiotic receipt. Multivariable negative binomial regression was used to analyze outcomes, controlling for clinically important patient characteristics. Results: Of 251 children included in this study, the median age was 4.2 years (IQR, 1.6–7.0); 52% were male, 40% were White, 54% were Hispanic, and 75% had government insurance. Antibiotics were prescribed in 26% of ILI encounters. There was no statistically significant association between antibiotic receipt and number missed class days (2.0 days [IQR, 1.0–4.0] vs 3.0 days [IQR, 1.0–5.0]; P = .08) or illness days (4.0 days [IQR, 3.0–7.0] vs 5.0 days [IQR, 3.0–7.0]; P = .13) after the initial ED visit. After adjusting for covariates, there was no significant difference in missed class days or illness days for patients prescribed antibiotics in relation to days sick before ED visit. The rates of missed class days and illness days were 87% and 30% greater, respectively, in patients with additional medical visits during the course of illness. Conclusions: Days sick prior to ED presentation and receipt of an antibiotic for ILI had no influence on child absenteeism or illness duration. However, children missed more class and received more antibiotics if they had multiple medical visits during an illness. Further study is needed on sociobehavioral factors leading to medicalization of children with minor illnesses and its impact on the unnecessary use of antibiotics.
Funding: None
Disclosures: None
C. difficile
In veteran outpatients, antibiotics remain significant risk factor for community-acquired Clostridiodes difficile infection
- Ukwen Akpoji, Brigid Wilson, Tayoot Chengsupanimit, Sunah Song, Taissa Bej, Robin Jump, Federico Perez
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s26-s27
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: An estimated 30% of antibiotic prescriptions in outpatient settings may be inappropriate. Antibiotic exposure increases an individual’s risk of Clostridioides difficile infection (CDI). To assess the prevalence of community-acquired CDI (CA-CDI) among patients without recent hospitalization and to examine the influence of outpatient antibiotic exposure on the risk of acquiring CA-CDI in this population, we examined a 2-year cohort of patients seen in primary care clinics at VA community-based outpatient clinics (CBOCs) associated with a large VA medical center. Methods: All primary care visits and nonvisit antibiotic prescriptions were identified in calendar years 2018–2019 as encounters of interest. Encounters occurring Results: We identified 84,787 patients with visits meeting our criteria. In this cohort, 3,533 patients were prescribed antibiotics at their encounter of whom 5 (0.14%) developed CA-CDI. Among the 81,254 patients who were not prescribed antibiotics, 15 (0.02%) developed CA-CDI, yielding an unadjusted CA-CDI odds ratio of 7.68 (95% CI, 2.50–19.82). p Conclusions: Although CA-CDI episodes were infrequent among VA outpatients with a CBOC visit in 2018–2019, the odds of CA-CDI were 7-fold greater in outpatients with antibiotic exposure than outpatients without antibiotic exposure. Antibiotic stewardship interventions that emphasize adverse events as a result of care provided in the outpatient setting, rather than as events limited to acute-care settings, may mitigate CDI risk.
Funding: This work was supported by the Merck Investigator Studies Program (MISP 59266 to F.P. and R.J.), and funds and facilities were provided by the Cleveland Geriatric Research Education and Clinical Center (GRECC) at the VA Northeast Ohio Healthcare System. The funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Disclosures:. All authors report no conflicts of interest relevant to this article. R.J. has received research funding from Pfizer; she has also participated in advisory boards for Pfizer and Merck.

Characteristics of antibiotic exposures for surgical procedures prior to Clostridioides difficile diagnosis—Minnesota, 2018
- Paige D’Heilly, Amanda Beaudoin, Davis Melin, Stacy Holzbauer
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s27
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background:Clostridioides difficile infection (CDI) is the leading cause of healthcare-associated diarrhea. Significant risk factors for CDI include antibiotic use and healthcare exposure. Antibiotics are often administered before, during and/or after surgery to prevent postsurgical infection. The contribution of surgery-related antibiotics to the overall CDI burden has not been well described, and assessment of the appropriateness of surgical antibiotic use is complicated by complex clinical guidelines. We have described surgical antibiotic prophylaxis history among adult with CDI in Minnesota in 2018. Method: The Minnesota Department of Health (MDH) performs 5-county active population- and laboratory-based CDI surveillance as a CDC Emerging Infections Program site. Incident CDI was defined as stool positive for C. difficile by toxin or molecular assay from a person aged ≥18 years with no positive test in the preceding 8 weeks. History of CDI was defined as having had a previous CDI episode in the 2009–2018 surveillance data set. Medical records were reviewed for 12 weeks prior to incident CDI test date to identify antibiotic prescriptions. Antibiotics with documented indication for surgical-site infection prevention or surgical prophylaxis were classified as “surgical antibiotic prophylaxis” (SPPX). SPPX type (eg, intraoperative, postoperative), appropriateness of SPPX, and clinical guideline adherence were not assessed. Results: During 2018, 812 incident CDIs were reported to MDH among 736 patients. SPPX preceded 84 (10.3%) cases, non-SPPX antibiotic use preceded 465 cases (57.3%), and 263 cases (32.4%) had no documented prior antibiotic use. The median age of incident CDIs with preceding SPPX was 68 years (IQR, 54–79.5). In 25 incident CDI cases with preceding SPPX (29.8%), there were no other antibiotic exposures. Among incident CDIs with preceding SPPX, 11 (13.1%) had >1 surgery event with SPPX. Prior CDI was identified for 13 (15.7%) with SPPX. Among 99 procedures with preceding SPPX, orthopedic surgeries (n = 27, 27.3%), gastrointestinal surgeries (n = 26, 26.3%), and cardiovascular surgeries (n = 22, 22.2%) were most common. In total 18 SPPX prescriptions (18.2%) originated in outpatient settings. SPPX drugs included cefazolin (n = 67, 67.7%), ceftriaxone (n = 7, 7.1%), ertapenem (n = 6, 6.1%), and clindamycin (n = 6, 6.1%). Median SPPX duration was 1 day (IQR, 1–2), and the median number days between surgery and specimen collection date was 19 (IQR, 7–49). Conclusions: Antibiotic stewardship programs should assess surgical prescribing, including in outpatient centers. Even short antibiotic duration for surgery could put patients at risk for CDI. More data are needed to evaluate the appropriateness of SPPX prescribing and to describe the impact of SPPX on CDI.
Funding: None
Disclosures: None
Clostridioides difficile: A diagnostic intervention
- Majd Alsoubani, Joshua Khuvis, Angie Rodday, Shira Doron
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s27
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background:Clostridioides difficile infection (CDI) is a leading cause of healthcare-associated infection and is associated with increased morbidity and mortality. Multiple strategies have been implemented to optimize the diagnostic accuracy of CDI testing algorithms. However, overdiagnosis of C. difficile colonization remains a challenge especially in the era of highly sensitive Nucleic acid amplification testing (NAAT). We implemented 2 interventions to reduce the rates of inappropriate C. difficile orders and tests. Methods: We performed a quasi-experimental retrospective study to examine the rates of all inpatient C. difficile test orders and results relative to 2 interventions between January 2018 and February 2021. We defined 3 periods: preintervention, after the first intervention, and after the second intervention. The first intervention, implemented May 2019, was a clinical decision support system (CDSS) tool to guide clinicians to order testing only if CDI criteria were met. The second intervention, implemented July 2020, was the requirement of mandatory antimicrobial team approval of PCR reflex testing for indeterminate toxin or antigen test results. This intervention included a discussion between clinicians and members of stewardship team prior to approval. To evaluate the impact of interventions on ordering appropriateness, chart review was conducted on a random subsample of 100 orders from each period. Hospital-onset CDI (HO-CDI) was calculated using CDC criteria. Results: In total, 3,004 C. difficile test orders were placed during the study period. The rates of reportable HO-CDI were significantly reduced by 57.1% (P = .003). We detected a significant reduction in the number of tests ordered over time from 11.6 to 7.51 per 1,000 patient days. (p Conclusions: CDSS tools target patients at high pretest probability of CDI. The restriction of PCR-reflex testing to the antimicrobial stewardship team is a novel effective measure to minimize the misdiagnosis of CDI. The incorporation of multiple strategies is necessary to improve the diagnostic accuracy of C. difficile testing.
Funding: None
Disclosures: None
Clostridioides difficile infection (CDI) treatment outcomes and recurrence factor at a pediatric hospital
- Martin Tuan Tran, Jasjit Singh, Wendi Gornick, Beth Huff, Negar Ashouri
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s28
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: CDI is the single most common cause of nosocomial diarrhea in both adults and children. Available data regarding treatment outcomes in hospitalized children remain limited. CDI recurrence in children has been reported in 20%–30% of cases. Consensus regarding the best testing method for CDI is lacking. The 2018 IDSA guideline recommends a multistep algorithm with detection of glutamate dehydrogenase antigen plus toxin, followed by detection of toxigenic C. difficle with nucleic acid amplification test (NAAT) if results are discordant. Methods: We included patients aged 1–26 years admitted from July 2020 through June 2021 with CDI symptoms and positive toxin or NAAT. Healthcare facility-onset CDI (HO-CDI) was defined as positive specimen collected >3 days after admission. Community-onset CDI (CO-CDI) was defined as positive specimen collected ≤3 days after admission. Community-onset healthcare facility-associated CDI (CO-HCFA-CDI) was defined as positive specimen from a patient who was discharged from the facility ≤4 weeks prior. Recurrence was defined as an episode of CDI occurring within 60 days after onset of a previous infection. Results: Mean age of the 63 patients meeting inclusion criteria was 11.2 years (range, 1–21 years). Most patients (n = 37; 58.7%) were male, tested negative for C. difficile toxins (n = 39; 61.9%), and had mild-to-moderate disease (n = 61; 96.8%). Patients with immunocompromising conditions were common, including malignancy (n = 38; 60.3%), inflammatory bowel disorder (n = 8; 12.7%), and history of solid organ transplant (n = 5; 7.9%). Previously healthy without chronic medical conditions were uncommon (n = 4; 6.3%). CO-CDI was most common (n = 26; 41.3%) followed by HO-CDI (n = 23; 36.5%). Also, 34 patients (53.9%) were exposed to antibiotics within the previous 30 days, 16 (47.0%) of whom received 2 or more antibiotics. Sulfamethoxazole–trimethoprim was the most prescribed agent (13; 38%), most (12; 92.3%) as prophylaxis for Pneumocystis jirovecii pneumonia. Furthermore, 42 patients (66.7%) were receiving gastric acid suppressant agents. Laxatives were given to 14 patients (22.2%) within 72 hours of testing, despite electronic reminders. Most were treated with oral vancomycin (n = 46; 73.0%). In addition, 5 patients (7.9%) did not receive CDI treatment at the discretion of the treating physician; all were toxin negative. CDI was cured in 58 patients (92.1%) with only 5 (7.9%) experiencing recurrence infection. Patients testing positive for C. difficile toxin were more likely to experience infection recurrence compared to those with a negative toxin screen: 4 of 24 (16.7%) versus 1 of 39 (2.6%) (P = .044). Conclusions: Most patients with CDI were treated with oral vancomycin at our institution. We observed significantly lower rate of recurrence than previously reported. Toxin-positive patients experienced higher recurrence rate. Prospective studies are needed to confirm our findings.
Funding: None
Disclosures: None
Role of diagnostic stewardship in reducing healthcare facility-onset Clostridioides difficile infections
- Anita Shallal, Medha Cherabuddi, Lance Podsiad, Christopher Gortat, Clare Shanahan, Jessica Kirkpatrick, Eman Chami, Stephanie Schuldt, Tarlisha Holsey, Martin Levesque, George Alangaden, Geehan Suleyman
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s28-s29
-
- Article
-
- You have access Access
- Open access
- Export citation
-
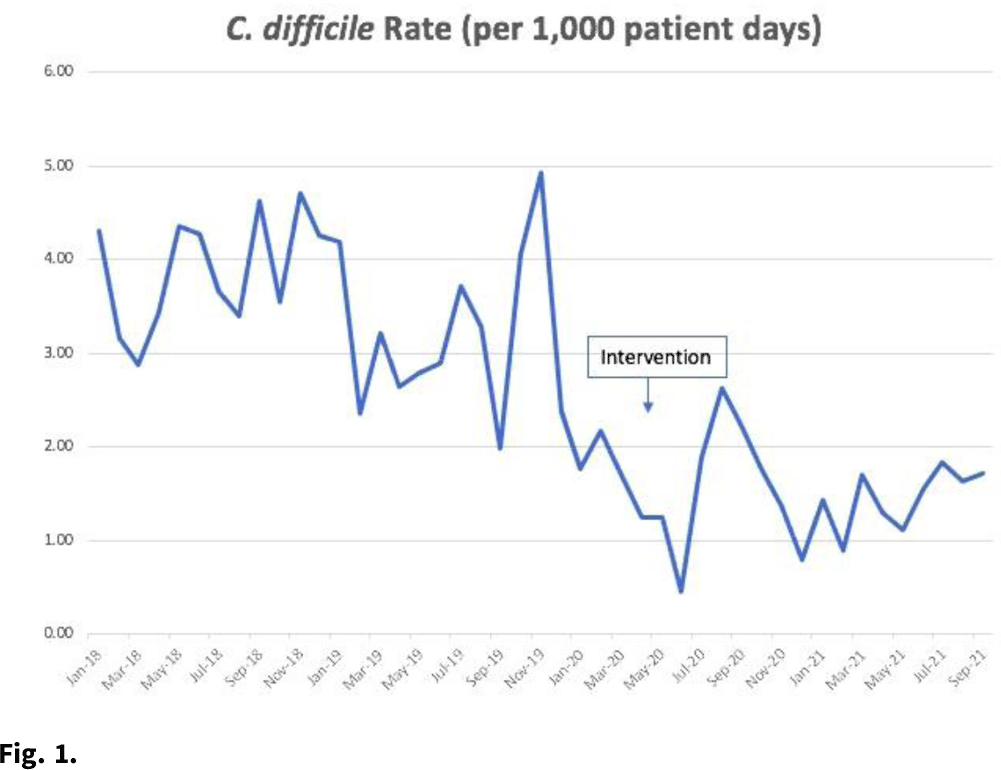
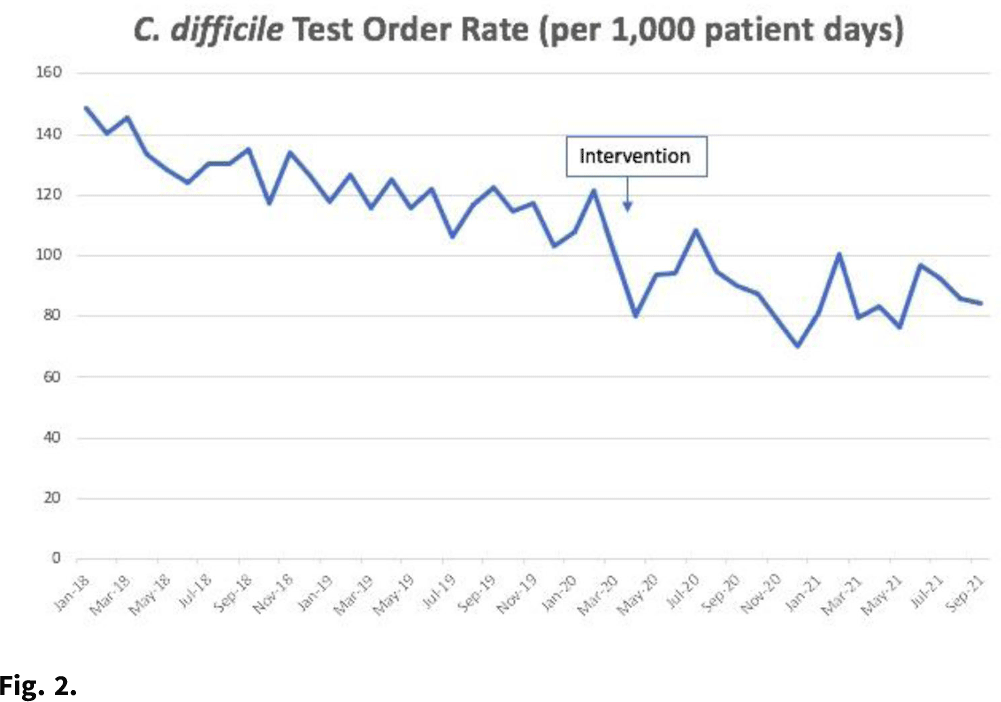
Background:Clostridioides difficile infection (CDI) is the most common healthcare-associated infection (HAI) in the United States. Healthcare facility-onset (HO) CDI reporting is a laboratory-identified (LabID) event and does not rely on symptoms. Inappropriate testing can lead to overdiagnosis in patients who are colonized, especially in those receiving promotility agents. Approximately 45% of HO-CDI cases at our institution occurred in the setting of laxative use in 2019. We assessed the effectiveness of an electronic medical record (EMR) “hard stop” in reducing inappropriate CDI testing and its impact on HO-CDI rates. Methods: We conducted a pre–post quasi-experimental retrospective study comparing test order rates per 1,000 patient days, CDI rate per 1,000 patient days, and standardized infection ratio (SIR) in the preintervention period (January 2018–December 2019) to the intervention period (April 2020–September 2021), at a 5-hospital healthcare system in southeastern Michigan. In February 2020, we implemented a hard stop in Epic that was triggered >3 days after admission for the following criteria: patients <1 year of age; repeated testing within 7 days, and receipt of promotility agents within 48 hours. After discontinuing the promotility agents for at least 48 hours, providers were allowed to place an order if diarrhea persisted. The medical director of infection prevention and control or designee had the ability to override the hard stop when deemed necessary after reviewing the case upon provider request. All orders expired after 24 hours if a specimen was not collected. We retrospectively reviewed the number of overrides after the intervention to determine the positivity rate. Results: Our CDI rates per 1,000 patient days were 3.21 in the preintervention period and 1.48 in the postintervention period, a 54% reduction (Fig. 1). The test order rates were 119.4 in the preintervention period and 87.7 in the postintervention period, a 26.5% reduction (Fig. 2). The SIR decreased from 0.542 in the preintervention period to 0.361 in the postintervention period, a 33% reduction (95% CI, 0.54–0.82; P = .0001). After the intervention, 299 patients had an override. Of these, samples from 218 patients (72.9%) were negative, 50 orders (16.7%) were cancelled, and 28 samples (9%) were positive. Conclusions: Diagnostic stewardship, utilizing an electronic hard stop, was effective in reducing inappropriate C. difficile testing in the setting of promotility agents without delaying diagnosis of HO-CDI. This strategy combined with standard best practices can significantly reduce HO-CDI rates.
Funding: None
Disclosures: None
Prospective audit and feedback of Clostridioides difficile PCR at the time of ordering increases appropriateness of testing
- Daniel Tassone, Matthew Hitchcock, John Markley, Michael Stevens
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s29
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Over-testing for Clostridioides difficile infection outside acute diarrheal illness without a clear alternative cause can lead to inappropriate diagnosis and treatment with antibiotic therapy. Preanalytical interventions such as education, order restriction, and electronic order assistance are common but are limited in effectiveness. As an alternative approach, our antibiotic stewardship program (ASP) implemented prospective audit and feedback (PAF) on C. difficile PCR orders to reduce inappropriate testing.
Methods: The study was conducted at a 399-bed, tertiary-care, Veterans’ Affairs Medical Center and included adult inpatients and outpatients for whom C. difficile PCR testing was ordered. In the preintervention period from June through September 2019, the ASP was alerted to C. difficile PCR tests and collected data but did not intervene. From October 2019 to January 2020, the ASP performed real-time PAF at the time of ordering. Appropriateness of testing was determined based on whether there was a negative result in the prior 7 days and a 3-step review of clinical factors (Fig. 1). When possible, a direct conversation took place with the ordering provider. If not possible, a general note delineating appropriate clinical criteria for testing was generated. No PAF was done outside standard hours. The ASP recommended cancelling tests deemed inappropriate. Monthly test rates during the pre- and postintervention periods were compared using the Student t test with α = .05, and test appropriateness was compared using the χ2 test. All analyses were conducted using Microsoft Excel software. Results: During the preintervention period, a total of 418 tests were ordered (104.5 per month). This number decreased to 276 (69 per month) during the intervention period. (p Conclusions: Direct PAF at the time of C. difficile PCR ordering may increase test appropriateness and is associated with a reduction in overall testing, primarily by reducing the number of tests that are considered not appropriate on clinical grounds. PAF is effective but requires significant time investment by ASP staff and may not be a sustainable intervention over time.
Funding: None
Disclosures: None

Evaluation of the genomic epidemiology and transmission of Clostridioides difficile infection across a community
- Brenda Tesini, Samantha Taffner, Trupti Hatwar, Steven Gill, Ghinwa Dumyati, Nicole Pecora
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s29-s30
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background:Clostridioides difficile infection (CDI) is a major cause of morbidity and healthcare costs in the United States. The epidemiology of CDI has recently shifted, with healthcare-associated (HCA) CDI trending downward and community-associated (CA)-CDI becoming more prominent. The cause of this shift is not well understood but may be related to changing genomic epidemiology. We assessed C. difficile strains across a CDC Emerging Infections Program (EIP) site in Western New York, including strains from both HCA-CDI and CA-CDI cases to characterize predominating strains and putative transmission across epidemiological classifications and between index and recurrent cases. Methods: In total, 535 isolates of C. difficile were collected over a 6-month period in 2018 from the Monroe Country, New York, EIP site and were analyzed using whole-genome sequencing (WGS). Standard epidemiological definitions were used to classify cases as hospital onset (HO-CDI); community associated (CA-CDI); community onset, healthcare associated (CO-HCFA-CDI); or long-term care onset (LTCO-CDI). Recurrent cases were defined as those diagnosed within 8 weeks of an initial positive test. Multilocus sequence types (MLSTs) were assigned according to PUBMLST and single-nucleotide polymorphisms (SNPs) were determined using a modified CFSAN analytical pipeline. Cases resulting from putative transmission were defined as those separated by 0–1 core SNPs. Results: Of 535 isolates, 454 were from index and 81 were from recurrent cases. The index cases were comprised of CA-CDI (47.4%), CO-HCFA-CDI (24%), LTCO-CDI (8.1%), and HO-CDI (19.3%). Cases with recurrent disease mirrored the epidemiological distribution of the larger set. Common MLSTs included ST2 (12.3%), ST8 (10.5%), ST42 (7.9%), ST58 (4.9%), ST43 (4.5%), and ST11 (4.3%). The previously widespread epidemic strain, NAP1/ST1/RT027 accounted for Conclusions: The genomic epidemiology of C. difficile across this large community cohort demonstrated a diverse group of strain types that was similarly distributed across epidemiological classifications and between index and recurrent cases. SNP analysis indicated that direct transmission between cases was uncommon.
Funding: None
Disclosures: None
CLABSI
Peripheral intravascular catheter-associated bloodstream infection in the medical–surgical ICU
- Nancy Hogle, Patrick Burke, Thomas Fraser
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s30
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Prompt removal of unnecessary central venous catheters (CVC) may reduce central-line–associated bloodstream infection (CLABSI). Primary non–central-line–associated hospital-acquired bloodstream infection (BSI), including peripheral intravascular (PIV) catheter-associated bloodstream infection (PIVABSI) remains a problem. Hospitals use CLABSI surveillance data to measure patient safety, yet this measure alone fails to describe the burden of total intravascular device-related infection. We described non-CLABSI primary BSI due to PIV in our medical–surgical ICU population. Methods: Hospital-wide surveillance for primary hospital-acquired BSI, including CLABSI, was conducted in accordance with NHSN protocol. We measured PIV catheter days and central-line days using a database including nursing device documentation and patient census data to count the number of patients with 1 or more devices in place in each location, counted at the same time each day. By substituting the role of the CVC with short or midline PIV in NHSN CLABSI surveillance protocols, we performed surveillance for PIVABSI. We defined PIVABSI as a patient without CVC and either a short or midline catheter in place for >2 calendar days on the date of BSI. Patients with BSI and both CVC and PIV were counted as CLABSI. We compared CVC and PIV utilization and the incidence density of CLABSI and PIVABSI in 8 medical and surgical ICUs at our large teaching hospital. We used OpenEpi version 3.01 software to test the hypothesis that the incidence density of CLABSI would be significantly different from that of PIVABSI. Results: From January to September 2021, there were 16 CLABSIs and 12 primary non–central-line–associated hospital-acquired BSIs, all 12 were PIVABSIs. Of these 12, 8 had >1 PIV in place and none were midlines. There were 13,418 central-line days, 10,897 short and midline peripheral IV days, and 22,415 patient days, resulting in device utilization ratios of 0.60 and 0.49, respectively. The incidence density of CLABSI was 1.2 per 1,000 central-line days, although the incidence density of PIVABSI was 1.1 per 1,000 peripheral IV days (P = .84). There was no difference in pathogens between the 2 groups. Conclusions: PIVABSI represented more than one-third of the total primary hospital-acquired BSIs in our medical and surgical ICUs. Total BSI surveillance is feasible. Efforts to reduce CLABSI should be part of a broader strategy to decrease total hospital-acquired BSI from all vascular access devices.
Funding: None
Disclosures: None
Blood-culture ordering practices in patients with a central line at an academic medical center–Iowa, 2020
- Elias Kovoor, Takaaki Kobayashi, Lorinda Sheeler, Alexandra Trannel, William Etienne, Oluchi Abosi, Stephanie Holley, Mary Kukla, Angie Dains, Kyle Jenn, Holly Meacham, Beth Hanna, Alexandre Marra, Meredith Parsons, Bradley Ford, Melanie Wellington, Daniel Diekema, Jorge Salinas
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s30
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The IDSA has a clinical definition for catheter-related bloodstream infection (CRBSI) that requires ≥1 set of blood cultures from the catheter and ≥1 set from a peripheral vein. However, because blood cultures obtained from a central line may represent contamination rather than true infection, many institutions discourage blood cultures from central lines. We describe blood culture ordering practices in patients with a central line. Methods: The University of Iowa Hospitals & Clinics is an academic medical center with 860 hospital beds. We retrospectively collected data for blood cultures obtained from adult patients (aged ≥18 years) in the emergency department or an inpatient unit during 2020. We focused on the first blood cultures obtained during each admission because they are usually obtained before antibiotic initiation and are the most important opportunity to diagnose bacteremia. We classified blood-culture orders as follows: CRBSI workup, non-CRBSI sepsis workup, or incomplete workup. We defined CRBSI workup as ≥1 blood culture from a central line and ≥1 peripheral blood culture (IDSA guidelines). We defined non-CRBSI sepsis workup as ≥2 peripheral blood cultures without cultures from a central line because providers might have suspected secondary bacteremia rather than CRBSI. We defined incomplete workup as any order that did not meet the CRBSI or non-CRBSI sepsis workup. This occurred when only 1 peripheral culture was obtained or when ≥1 central-line culture was obtained without peripheral cultures. Results: We included 1,150 patient admissions with 4,071 blood cultures. In total, 349 patient admissions with blood culture orders (30.4%) met CRBSI workup. 62.8% were deemed non-CRBSI sepsis workup, and 6.9% were deemed an incomplete workup. Stratified by location, ICUs had the highest percentage of orders with incomplete workups (8.8%), followed by wards (7.2%) and the emergency department (5.1%). In total, 204 patient admissions had ≥1 positive blood culture (17.7%). The most frequently isolated organisms were Staphylococcus epidermidis (n = 33, 16.2%), Staphylococcus aureus (n = 16, 7.8%), and Escherichia coli (n = 15, 7.4%) Conclusions: Analysis of blood culture data allowed us to identify units at our institute that were underperforming in terms of ordering the necessary blood cultures to diagnose CRBSI. Being familiar with CRBSI guidelines as well as decreasing inappropriate ordering will help lead to early and proper diagnosis of CRBSI which can reduce its morbidity, mortality, and cost.
Funding: None
Disclosures: None
The evaluation of central-line–associated bloodstream infection (CLABSI) preventability at an academic institution
- Leon Hsueh, Daniel Uslan, Annabelle De St. Maurice
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s30-s31
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: In 2008, the hospital-acquired conditions (HACs) initiative labeled central-line–associated bloodstream infections (CLABSIs) as preventable “never events” that could no longer be reimbursed by Medicare. However, some patients have inherent unpreventable etiologies for bacteremia, such as obstructive biliary malignancies. We assessed the number of CLABSIs that were reasonably preventable. Methods: We examined all CLABSI cases at 2 academic medical centers over a 2-year period (2019–2021). We established 3 categories of CLABSIs: (1) preventable CLABSI (pCLABSI); (2) end-of-life CLABSI (EOL-CLABSI), which were CLABSIs that were caused by underlying disease processes in patients who were nearing the end of their lives due to a debilitating comorbidity; and (3) definition-based (dCLABSI), which met NHSN criteria for a CLABSI but, based on the pathogen and the clinical situation, likely occurred as a consequence of a patient’s comorbidities. Two experienced infectious diseases physicians (D.U. and A.S.M.) reviewed the charts of each patient with a CLABSI and, based on expert opinion, determined the category for each CLABSI. Results: Over the 2-year period, 147 CLABSIs were identified among the 2 hospitals, 66 (44.9%) of which occurred in an ICU. Most CLABSIs were pCLABSIs, making up 99 CLABSIs (67.3%). In comparison, 20 cases were categorized EOL-CLABSIs (13.6%), although 26 cases were dCLABSIs (17.7%), and 2 cases could not be classified. There was no difference in the distribution of CLABSI types in an ICU versus a non-ICU setting (χ2P = .265). However, we detected microbiologic differences between pCLABSIs, EOL-CLABSIs, and dCLABSIs (χ2P < .001), with gram-positive cocci making up the large majority of pCLABSIs (62.6%), followed by Candida spp (24.2%). Gram-negative bacilli (GNR) made up 11.1% of pCLABSIs. In comparison, GNRs were more prevalent in EOL-CLABSIs and dCLABSIs, making up 30.0% and 38.5% of each CLABSI type, respectively. Conclusions: Two-thirds of CLABSIs were deemed preventable. Central lines are important for managing critically ill patients, many of whom have inherent risk factors for bloodstream infections. EOL-CLABSIs highlight the potential for early care discussions to avoid CLABSIs at the end of a patient’s life and to avoid unnecessary blood cultures for patients on comfort care. Additionally, the pCLABSI distinction allows hospital epidemiology teams to focus on the CLABSI cases that can realistically be prevented with appropriate central-line care, techniques, and hand hygiene. Creating these categories allows hospital systems to use more targeted approaches for improving CLABSI rates.
Funding: None
Disclosures: None
The effectiveness of a dedicated central venous access care team to prevent catheter-related bloodstream infections
- Fortune Charles Fil de Lara, Maria Jesusa Mano, Karl Evans Henson, Jia An Bello, Cybele Lara Abad
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s31
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Catheter-related bloodstream infection (CRBSI) rates remain high despite the use of an insertion bundle. We hypothesized that line care and maintenance by a dedicated team would help decrease CRBSI rates. This study was conducted in The Medical City (TMC), is a 526-bed, private, tertiary-care center in Pasig City, Philippines. Methods: All adult hospitalized patients from October 1, 2020, to October 31, 2021, with a newly inserted temporary central venous catheter (CVC) were eligible for inclusion. CRBSI rates before the intervention (October 2019 to March 2020) and after the intervention (April to October 2021) were compared. The intervention arm consisted of a dedicated central venous access team (CVAT) who provided education and performed daily line care and dressing changes per protocol. A series of χ2 and Wilcoxon rank-sum tests were performed to compare characteristics between exposure groups. Incidence rates of CRBSI before and after the intervention were compared using an incidence rate ratio approach. Results: In total, 209 CVCs were enrolled in the study, with 103 CVCs (49.28%) in the preintervention arm and 106 CVCs (50.72%) in the postintervention arm. Baseline patient characteristics were similar. CRBSIs were more frequent in the preintervention arm than the postintervention arm (39 of 103 vs 28 of 106; P = .08). The CRBSI incidence density rate was higher in the preintervention arm than the postintervention arm, but the difference was not statistically significant (37.46 per 1,000 patient days vs 25.97 per 1,000 patient days; P = .14). Median time to CRBSI was similar in both groups (9 vs 8 days). Conclusions: Baseline CRBSI rates were high and risk of infection increased by day 8 after line insertion. We detected a decreasing trend in rates of CRBSI with a dedicated CVAT, but multiple interventions are likely needed to influence overall rates.
Funding: None
Disclosures: None

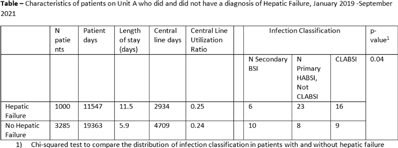
Hospital-acquired bloodstream infections in patients with and without hepatic failure
- Jordan Bosco, Patrick Burke, Francisco Marco Canosa, Stephen Wilson, Steven Gordon, Thomas Fraser
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s31-s32
-
- Article
-
- You have access Access
- Open access
- Export citation
-
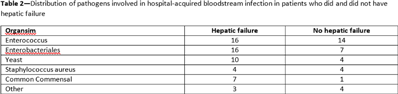
Background: The NHSN parameter estimate for predicted number of central-line–associated bloodstream infection (CLABSI) is the same for gastroenterology wards as other specialty wards, such as behavioral health and gerontology. We conducted this study to contribute to the body of knowledge surrounding the risk for hospital-acquired bloodstream infection (HABSI) in patients with and without hepatic failure. The Cleveland Clinic is a 1,200-bed, multispecialty hospital with a solid-organ transplant service. Patients with hepatic failure who do not require critical care are housed on 36-bed unit A. On unit A, 43% of patients are under hepatology or gastroenterology service, although 51% of patients are under general internal medicine. Overall, unit A has a high incidence of HABSI. Methods: Surveillance for HABSI and CLABSI is performed at the Cleveland Clinic per NHSN protocol. All patients with a midnight stay on unit A from January 2019 through September 2021 were dichotomized as having hepatic failure (yes or no) if they ever received the International Classification of Diseases Tenth Revision code for “hepatic failure, not elsewhere classified.” We joined the diagnostic code to patient days and central-line-days databases and summarized the data using Microsoft Excel software. We stratified the number of patients, patient days, device days, infection classification, and hospital length of stay by whether the patient had hepatic failure, and we compared the incidence of HABSI and CLABSI between the 2 groups using OpenEpi version 3.01 software. Results: We identified 72 HABSIs among 4,285 patients who stayed on unit A for 30,910 patient days during the study period. The incidences of HABSI in patients with and without hepatic failure were 39.0 and 13.9 per 10,000 patient days, respectively (P < .001). The incidence of CLABSI was 5.4 and 1.9 per 1,000 line days, respectively (P = .01). Patients with hepatic failure stayed longer (11.5 vs 5.9 days), yet the central-line utilization ratios were not substantially different (0.25 vs 0.24). Enterococcus was the most common pathogen involved in CLABSI in both groups (Table 2). Conclusions: Patients with hepatic failure experienced CLABSI more frequently than patients without hepatic failure, stayed longer in the hospital, and were less likely have HABSI attributed to another primary focus of infection according to NHSN definitions. Although hepatic failure may be among the most severe conditions among patients in a gastroenterology ward, we have demonstrated that these units house a population uniquely susceptible to HABSI and CLABSI.
Funding: None
Disclosures: None