Poster Presentation - Poster Presentation
Respiratory Viruses Other than SARS-CoV-2
Relevance of RSV in hospitalized adults and the need for continued testing
- Katherine Miller, Arnold Monto, H. Keipp Talbot, Manjusha Gaglani, Tresa McNeal, Fernanda Silveira, Richard Zimmerman, Donald Middleton, Shekhar Ghamande, Kempapura Murthy, Lindsay Kim, Jill Ferdinands, Manish Patel, Emily Martin
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s62
-
- Article
-
- You have access Access
- Open access
- Export citation
-
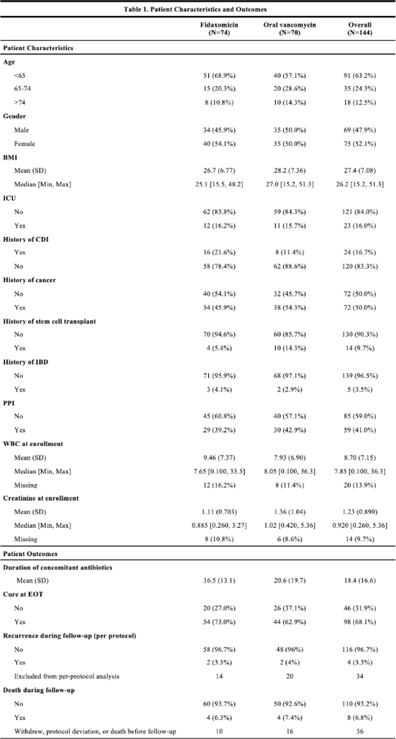
Background: RSV is underrecognized in hospitalized adults. A better understanding of RSV in this population could help prioritize targeted viral-testing resources. Hospitalization and in-hospital outcomes are widely accepted as markers of clinical severity with respect to acute respiratory illness (ARI). We compared characteristics and clinical outcomes between adults hospitalized with ARI from October 2016 through May 2019. Methods: All hospitalized adults (≥ 18 years) who met a standardized case definition of ARI were prospectively enrolled across 3 respiratory seasons from 9 hospitals participating in the US Hospitalized Adult Influenza Vaccine Effectiveness Network (HAIVEN). Demographic data were collected during enrollment interviews, and electronic medical records (EMRs) were reviewed to extract comorbidity data. Throat and nasal swabs collected at enrollment were tested for ARI pathogens using real-time PCR assays at respective HAIVEN research laboratory sites. Characteristics and clinical outcomes of participants were compared using χ2 or nonparametric tests where appropriate. Multivariable logistic regression models were used to test associations between infection status, characteristics, and clinical outcomes, adjusting for age, sex, race, Charlson comorbidity index (CCI), body mass index (BMI), site, season, and days to admission. Results: In total, 10,311 adults were included, 22.3% (n = 2,300) were aged 18–49 years, 33.2% (n = 3,423) were aged 50–64 years, and 44.5% (n = 4,588) were aged ≥65 years. Moreover, 6% of adults tested positive for RSV (n = 622), 18.8% positive for influenza (n = 1,940), and 75.1% negative for both (n = 7,749). Obesity and age ≥65 years were significantly associated with RSV detection when compared with participants negative for both RSV and influenza. Patients aged 18–49 years and ≥65 years with RSV had significantly higher median CCI scores compared to patients with influenza (Fig. 1.). The proportion of adults with CHF or COPD was significantly (p-value Conclusions: Severe RSV illness may differ from severe influenza illness, and those infected with RSV may have different characteristics than those infected with influenza. Hospitalized adults with RSV infection were more likely to have underlying cardiopulmonary comorbidities and higher CCI scores as well as experience an extended length of hospital stay and need for mechanical ventilation. These data highlight the importance of retaining testing for RSV in older adults hospitalized with ARI.
Funding: None
Disclosures: None
SSI
Feasibility and acceptability of intranasal povidone iodine decolonization among orthopedic trauma surgery patients
- Marin Schweizer, Loreen Herwaldt, Linda Boyken, Jean Pottinger, Rachel Quinn, Daniel Diekema, Fiona Armstrong Pavlik, Melissa Ward, Poorani Sekar, Michael Willey
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s62-s63
-
- Article
-
- You have access Access
- Open access
- Export citation
-
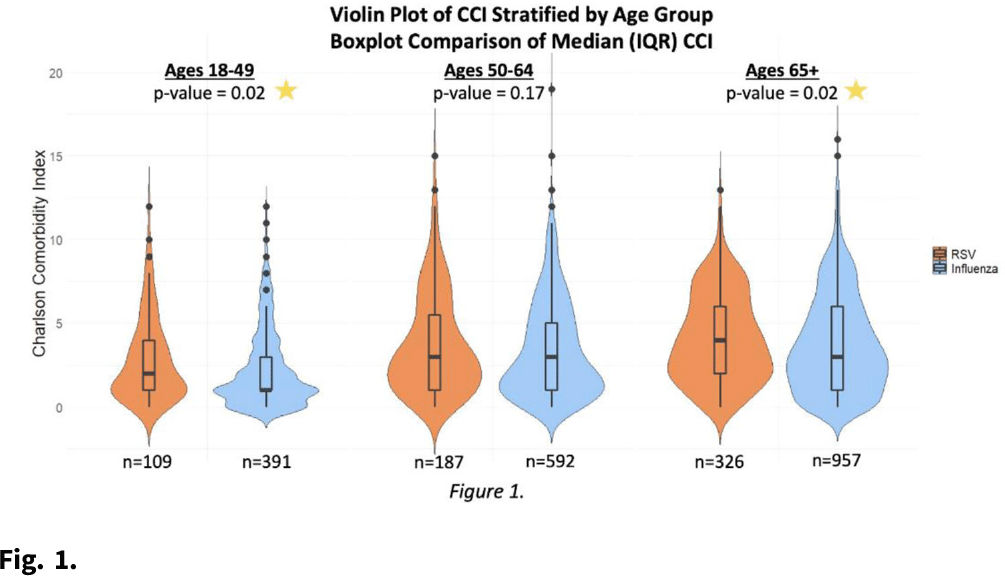
Background: Nasal decolonization significantly decreases the incidence of Staphylococcus aureus surgical-site infections (SSIs). Patient adherence with self-administration of a decolonization ointment (ie, mupirocin) is low, especially among patients having urgent surgery. Povidone-iodine decolonization may overcome patient adherence challenges because povidone-iodine needs to be applied only on the day of surgery. We assessed the effectiveness and acceptability of povidone-iodine decolonization given on the day of surgery among patients having orthopedic trauma surgery. Methods: Adult patients who underwent operative fixation of traumatic lower extremity fractures were consented to receive 10% intranasal povidone-iodine solution. Povidone-iodine was applied ~1 hour before surgical incision and was reapplied the evening after surgery. Patients were tested for S. aureus nasal colonization before surgery, the evening after surgery (before povidone-iodine reapplication), and the day after surgery. Swabs were inoculated into Dey-Engley neutralizer and processed in a vortexer. A series of dilutions were performed and plated on mannitol salt agar plates. S. aureus cultures were quantitatively assessed to determine the reduction in S. aureus after povidone-iodine use. Reductions in S. aureus nasal growth were evaluated using the Skillings-Mack test. SSIs manifesting within 30 and 90 days of surgery were identified using NHSN definitions. A survey was administered the morning after surgery to determine the acceptability of intranasal povidone-iodine. Results: In total, 51 patients participated in this pilot study between February 2020 and June 2021. Nasal samples from 12 participants (23.5%) grew S. aureus. The S. aureus concentration decreased significantly across the time points (P = .03) (Fig. 1). No SSIs were identified within 30 days of surgery. One SSI occurred within 90 days of surgery; this patient did not carry S. aureus, and cultures from the infected site were negative. Also, 31% of patients reported at least 1 mild side effect while using povidone-iodine: dripping (n = 7), itching (n = 6), dryness (n = 4), stinging (n = 4), staining (n = 3), unpleasant taste (n = 3), runny nose (n = 2), burning (n = 1), sneezing (n = 1), sore throat (n = 1), tickling (n = 1), and/or cough (n = 1). Also, 86% of patients stated that povidone-iodine felt neutral, pleasant, or very pleasant, and only 14% stated that it felt unpleasant or very unpleasant. Discussion: In this pilot study, 2 applications of nasal povidone-iodine on the day of surgery were acceptable for patients, and this protocol significantly reduced S. aureus concentration in nares of patients. Future large clinical trials should evaluate whether this 2-application regimen of povidone-iodine significantly decreases rates of SSI among orthopedic trauma surgery patients.
Funding: PDI Healthcare
Disclosures: None
Implementation of surgical site infection surveillance in 16 health facilities in Sierra Leone
- Rugiatu Z. Kamara, Monique Foster, Jamine Weiss, Christiana Conteh
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s63
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Surgical site infections (SSIs) are associated with increased healthcare costs, antibiotic resistance, morbidity, and mortality. In low- and middle-income countries (LMICs), SSIs account for most healthcare-acquired infections (HAIs). In Africa, up to 20% of women who undergo a caesarean section develop a wound infection. Surveillance has been shown to be an essential component in the overall strategy to reduce SSIs. Methods: Surgical site infection surveillance is being implemented in 16 health facilities in Sierra Leone, with at least 1 from each of the 5 US Census regions: Eastern, Western, Northern, Northwestern, and Southern. These health facilities were selected based on the availability of a dedicated infection prevention and control (IPC) focal person. Women were observed for 30 days after caesarean section. A standardized surgical safety and surveillance checklist including case definitions and observable criteria (eg, purulent drainage, wound abscess, or intentional reopening) was used. Clinical staff were trained to collect data and to conduct in-person and phone interviews with patients on days 3, 7, and 30 after caesarean section. Results: From March 2021 to July 2021, a total of 2,529 women had caesarean sections in 15 health facilities; most occurred in the Northern region (785 of 2,529). Among these 2,529 women, 1,522 (60%) had an SSI surveillance checklist started, and of those 1,522, 632 (42%) had a completed checklist. Health facilities in most of the rural regions, (Eastern, Northwestern, and Southern) had no completed checklists. The overall SSI rate for the 15 health facilities was 3% (70 of 2,529). The Southern region had the highest SSI rate at 50% (35 of 70), but the Western region did not report any SSIs. Of the 70 cases, 49 (70%) were identified through active inpatient surveillance and 21 (30%) were identified through postdischarge surveillance. Conclusions: One of the priorities of Sierra Leone’s National IPC Action Plan is to establish HAI surveillance. Surgical site surveillance is an essential component of HAI surveillance and leads to timely identification so infections can be treated quickly. This study was limited by inadequate data collection and patients lost to follow-up after discharge. However, this study illustrates that surveillance leads to the diagnosis of most SSI cases after caesarean section while patients are still hospitalized. Simple yet effective SSI surveillance can be conducted in LMICs to identify and ultimately treat SSI after caesarean section. More support is needed in rural and smaller facilities for better implementation of SSI surveillance in Sierra Leone.
Funding: None
Disclosures: None
Surveillance/Public Health
Increases in methicillin-sensitive Staphylococcus aureus bloodstream infection incidence, 2016–2019
- Kelly Jackson, Joelle Nadle, Susan Ray, Ruth Lynfield, Ghinwa Dumyati, Marissa Tracy, William Schaffner, David Ham, Isaac See
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s63-s64
-
- Article
-
- You have access Access
- Open access
- Export citation
-
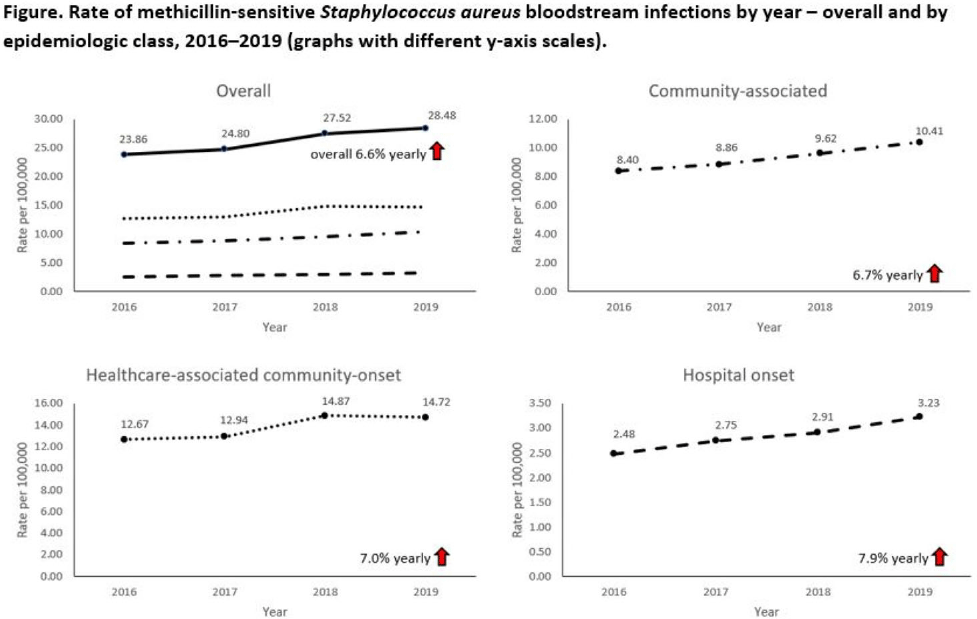
Background: Incidence of methicillin-sensitive Staphylococcus aureus (MSSA) bloodstream infections (BSIs) in the United States during 2012–2017 has been reported to have been stable for hospital-onset BSIs and to have increased 3.9% per year for community-onset BSIs. We sought to determine whether these trends continued in more recent years and whether there were further differences within subgroups of community-onset BSIs. Methods: We analyzed CDC Emerging Infections Program active, population- and laboratory-based surveillance data during 2016–2019 for MSSA BSIs from 8 counties in 5 states. BSI cases were defined as isolation of MSSA from blood in a surveillance area resident. Cases were considered hospital onset (HO) if culture was obtained >3 days after hospital admission and healthcare-associated community-onset (HACO) if culture was obtained on or after day 3 of hospitalization and was associated with dialysis, hospitalization, surgery, or long-term care facility residence within 1 year prior or if a central venous catheter was present ≤2 days prior. Cases were otherwise considered community-associated (CA). Annual rates per 100,000 census population were calculated for each epidemiologic classification; rates of HACO cases among chronic dialysis patients per 100,000 dialysis patients were calculated using US Renal Data System data. Annual increases were modeled using negative binomial or Poisson regression and accounting for changes in the overall population age group, and sex. Descriptive analyses were performed. Results: Overall, 8,344 MSSA BSI cases were reported. From 2016–2019 total MSSA BSI rates increased from 23.9 per 100,000 to 28.5 per 100,000 (6.6% per year; P < .01). MSSA BSI rates also increased significantly among all epidemiologic classes. HO cases increased from 2.5 per 100,000 to 3.2 per 100,000 (7.9% per year; P = .01). HACO cases increased from 12.7 per 100,000 to 14.7 per 100,000 (7.0% per year; P = .01). CA cases increased from 8.4 per 100,000 to 10.4 per 100,000 (6.7% per year; P < .01) (Fig. 1). Significant increases in MSSA BSI rates were also observed for nondialysis HACO cases (9.3 per 100,000 to 11.1 per 100,000; 7.8% per year; P < .01) but not dialysis HACO cases (1,823.2 per 100,000 to 1,857.4 per 100,000; 1.4% per year; P = .59). Healthcare risk factors for HACO cases were hospitalization in the previous year (82%), surgery (31%), dialysis (27%), and long-term care facility residence (19%). Conclusions: MSSA BSI rates increased from 2016–2019 overall, among all epidemiologic classes, and among nondialysis HACO cases. Efforts to prevent MSSA BSIs among individuals with healthcare risk factors, particularly those related to hospitalization, might have an impact on MSSA BSI rates.
Funding: None
Disclosures: None
Reduction in outpatient antibiotic utilization: An unintended benefit of the COVID-19 pandemic?
- Satoshi Kakiuchi, Eli Perencevich, Daniel Livorsi, Michihiko Goto
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s64
-
- Article
-
- You have access Access
- Open access
- Export citation
-
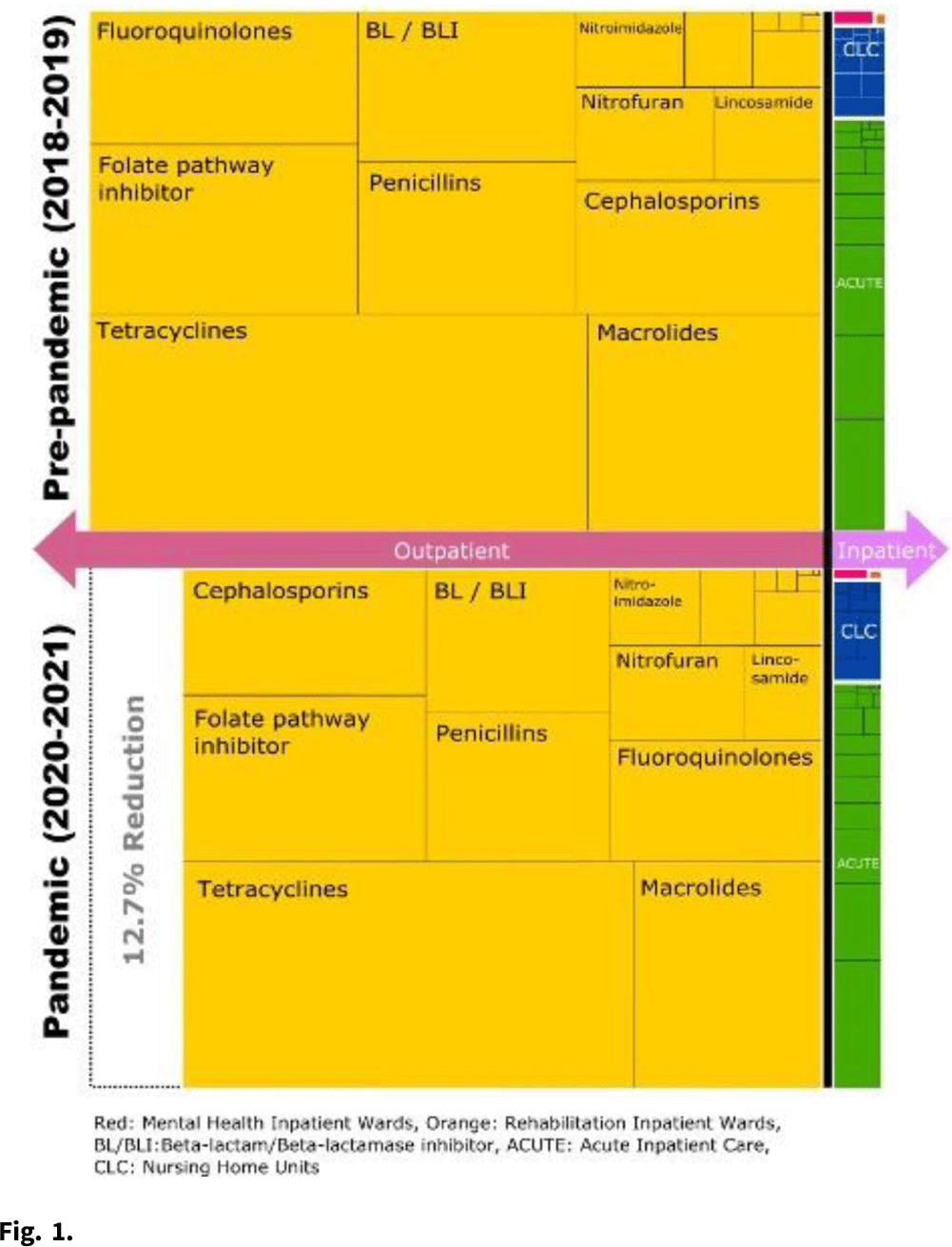
Background: The COVID-19 pandemic heavily affected healthcare delivery systems in the United States. However, little is known about its impact on overall antimicrobial consumption, especially in outpatient settings. We assessed the impact of the COVID-19 pandemic on antimicrobial consumption in both outpatient and inpatient (acute-care, long-term care, and mental health) settings in the Veterans’ Health Administration (VHA) during the 2 years before and after the start of the pandemic. Methods: We conducted a retrospective study for all patients who received care within the VHA from January 2018 to December 2021. We used antibiotic days as the primary outcome measure (days of therapy for inpatient settings and dispensed days supply for outpatient settings), and we obtained data for antimicrobial consumption from the VHA Corporate Data Warehouse. Antibiotics were categorized into classes by the NHSN protocol and included only systemic agents (oral and parenteral). We defined 2018–2019 as the prepandemic period and 2020–2021 as the pandemic period. We compared the relative and absolute difference in antibiotic consumption between the 2 periods. Results: Across all periods, 8.3 million patients received care in the VHA, and an average of 28,709,680 antibiotic days were prescribed per year. Overall, 92.9% of all antibiotic days were outpatient and 7.1% were inpatient. Total antibiotic days during the pandemic period decreased by 12.4% compared to the prepandemic period (pandemic period: 53,613,840 and prepandemic period: 61,224,878). This reduction was primarily driven by reductions in outpatient settings (relative reduction: 12.7% and absolute reduction: 7,254,880 antibiotic days over 2 years), but antibiotic days in inpatient settings decreased more modestly (relative reduction: 8.4% and absolute reduction: 356,158 antibiotic days over 2 years) (Fig. 1). When frequently prescribed antimicrobials were categorized by classes, fluoroquinolones and lincosamides showed the largest decreases (fluoroquinolones: 29.2% reduction and lincosamides: 27.2% reduction). Tetracyclines and sulfamethoxazole–trimethoprim had the smallest reductions (5.2% and 11.2%, respectively). Conclusions: Compared to the prepandemic period, the pandemic was associated with a substantial reduction in overall antibiotic consumption, especially in outpatient settings, which accounted for 95% of the overall reduction despite being outside the domain of most traditional antibiotic stewardship programs. The impact of the pandemic was most modest in the use of tetracyclines and trimethoprim–sulfamethoxazole and was most prominent in the use of fluoroquinolones and lincosamides. Further studies are required to improve the causal inference between the COVID-19 pandemic and this reduction in antibiotic consumption, as well as its impact on patient outcomes.
Funding: None
Disclosures: None
Mortality rates among non-Hispanic Black and White persons in carbapenemase-producing Enterobacterales, Tennessee, 2015–2019
- Erika Kirtz, Rany Octaria, Carolyn Stover, Christopher Wilson, Allison Chan
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s64-s65
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Carbapenem-resistant Enterobacterales (CRE) are an urgent public health threat, particularly those that produce carbapenemase (CP-CRE). Certain risk factors associated with CRE acquisition have been well described, such as older age, indwelling devices, prior hospitalizations, and underlying conditions. However, data are limited regarding the association of CRE and health disparities, such as race and ethnicity. Published literature has consistently shown that minority groups, including but not limited to Non-Hispanic Black persons, have higher risks of developing adverse health outcomes. To better understand the impact of race and ethnicity in CP-CRE cases, we compared 1-year mortality rates among Non-Hispanic Blacks and Non-Hispanic Whites. Methods: CRE are reportable in Tennessee; isolates must be sent to the State Public Health Laboratory for carbapenemase detection and resistance mechanism testing. We linked 2015–2019 CP-CRE surveillance cases and laboratory data from our statewide surveillance system, the National Disease Surveillance System (NEDDS)-Base System, with the Tennessee Hospital Discharge Data System (HDDS) and vital records databases. Database linkage and data analyses were performed using SAS version 9.4 software. Results: Among 615 CP-CRE cases, the mean age was lower among non-Hispanic Blacks (59 years; SD, 16.6) compared to non-Hispanic Whites (mean, 65 years; SD, 15.7). Among 156 non-Hispanic Blacks with CP-CRE, 101 (64.7%) were nursing home residents, whereas 281 (71.1%) among the 395 non-Hispanic Whites were nursing home residents. Also, 64 Non-Hispanic Blacks (41%) died within 1 year of their first specimen collection date compared to 92 Non-Hispanic Whites (23.3%). Non-Hispanic Blacks with CP-CRE who died within 1 year had a mortality rate of 5.6 per 100,000 (95% CI, 4.21–6.94) Black population, which was 1.6 times higher than Non-Hispanic White persons at 3.5 per 100,000 (95% CI, 2.94–3.95; χ2P < .001) White population. Conclusions: Despite a lower mean age, non-Hispanic Black CP-CRE cases had a higher 1-year mortality rate than non-Hispanic Whites. Racial and ethnicity data often are missing or incomplete from surveillance data. Data linkages can be a valuable tool to gather additional clinical and demographic data that may be missing from public health surveillance data to improve our understanding of health disparities. Recognition of these health disparities among CRE can provide an opportunity for public health to create more targeted interventions and educational outreach.
Funding: None
Disclosures: None
Developing national benchmarks for antimicrobial resistance–NHSN, 2019
- Hsiu Wu, Erin O’Leary, Minn Soe, Jonathan Edwards
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s65
-
- Article
-
- You have access Access
- Open access
- Export citation
-
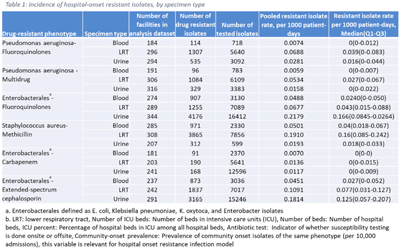
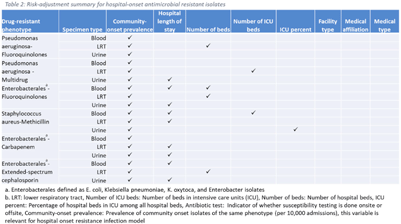
Background: The emergence and spread of drug-resistant pathogens continues to significantly impact patient safety and healthcare systems. Although antimicrobial susceptibility test (AST) results of clinical specimens are used by individual facilities for antimicrobial resistance surveillance, accurate tracking and benchmark comparison of a facility’s antimicrobial resistance using national data requires risk-adjusted methods to be more meaningful. The CDC NHSN Antimicrobial Resistance (AR) Option collects patient-level, deduplicated, isolate information, including AST results, for >20 organisms from cerebrospinal fluid, lower respiratory tract (LRT), blood, and urinary specimens. To provide risk-adjusted national benchmarks, we developed prediction models for incidence of hospital-onset isolates with antimicrobial resistance. Methods: We analyzed AST results of isolates reported through the NHSN AR Option for January through December 2019. Isolates from facilities that had >10% missing AST results for the organism-drug combinations or from hospitals that used outdated breakpoints were excluded. We assessed associations between facility-level factors and incidence rates of hospital-onset (specimen collected 3 days or more after hospital admission) isolates of specific drug-resistant phenotypes from blood, LRT, and urinary specimens. Factors included number of beds, length of stay, and prevalence of community onset isolates of the same phenotype. Drug-resistant phenotypes assessed included methicillin-resistant Staphylococcus aureus (MRSA), multidrug-resistant (MDR) Pseudomonas aeruginosa, carbapenem-resistant Enterobacterales (CRE), fluoroquinolone-resistant Pseudomonas aeruginosa, fluoroquinolone-resistant Enterobacterales, and extended-spectrum cephalosporin-resistant Enterobacterales. Isolates of different phenotypes and from different specimen sources were modeled separately. Negative binomial regression was used to evaluate the factors associated with antimicrobial resistance incidence. Variable entry into the models is based on significance level P Among the models, 1 for each drug-resistant phenotype-specimen type combination, the number of isolates with AST results ranged from 718 (Pseudomonas aeruginosa–fluoroquinolones, blood) to 16,412 (Enterobacterales–fluoroquinolones, urine). The pooled incidence rate was highest for fluoroquinolone-resistant Enterobacterales in urinary specimens (0.2179 isolates per 1,000 patient days) among all phenotype-specimen combinations evaluated (Table 1). The incidence of drug-resistant isolates was consistently associated with community-onset prevalence across models evaluated. Other associated factors varied across phenotype-specimen combinations (Table 2). Conclusions: We developed statistical models to predict facility-level incidence rates of hospital-onset antimicrobial resistant isolates based on community-onset drug-resistant prevalence and facility characteristics. These models will enable facilities to compare antimicrobial resistance rates to the national benchmarks and therefore to inform their antimicrobial stewardship and infection prevention efforts.
Funding: None
Disclosures: None
Findings from healthcare-associated infections data validation attestation in California general acute-care hospitals
- Nadia Barahmani, Andrea Parriott, Erin Epson, Genie Tang, N. Neely Kazerouni
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s65-s66
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Accurate and complete hospital healthcare-associated infection (HAI) data are essential to inform facility-level HAI prevention efforts and to ensure the validity and reliability of annual public reports. We implemented a validation attestation survey to assess and improve the HAI data reported by California hospitals via NHSN. Methods: The California Department of Public Health (CDPH) HAI Program invited all 401 general acute-care hospitals in California to participate in an annual HAI validation attestation survey in 2021. The survey was designed to be completed by the person with primary responsibility for HAI surveillance and reporting consistent with NHSN protocols and California laws. Survey questions addressed HAI reporting knowledge and practices and surgical procedures performed, and they included 3 hypothetical scenarios evaluating hospital application of HAI surveillance, decision making, and reporting methods. Results: We received responses from 345 hospitals (86%). For the 3 hypothetical scenarios, 171 hospitals (49.6%) correctly answered all 3 questions, 110 hospitals (31.9%) answered 2 questions correctly, 52 (15.1%) hospitals answered 1 question correctly, and 12 hospitals (3.5%) answered zero questions correctly. We did not detect a statistically significant association between facility type (ie, acute-care hospital, critical access hospital, long-term acute-care hospital, or rehabilitation hospital or unit) and the probability of getting all questions correct (Fisher exact P = .42). Of the 303 hospitals (88.0%) that perform at least 1 of the 28 surgical procedures reportable in California, 269 (88.8%) apply CDPH-recommended postoperative ICD-10 diagnosis flag codes to identify records that might indicate a possible surgical site infection (SSI). Moreover, ~289 (84.0%) hospitals confirmed that someone at their facility reviews CDPH quality assurance–quality control reports to verify the accuracy and completeness of their hospital’s reported HAI data. In 321 hospitals (93.0%) decisions about which infections are reported to NHSN are made solely by the infection preventionists or hospital epidemiologists, who are thoroughly familiar and follow NHSN protocol, definitions, and criteria. Conclusions: Most hospitals reported following best practices for evaluating records for SSIs; however, only half responded correctly to all 3 hypothetical scenarios. Our results highlight the need for ongoing education on HAI surveillance, decision making and reporting methods, and external HAI data validation in hospitals. This survey could serve as a model for other states that work with hospitals to improve HAI surveillance data and to ensure the integrity of public reports. Future research will link the results of this survey to NHSN validation audits.
Funding: None
Disclosures: None
Lessons learned: Characteristics of first-year COVID-19 hospital outbreaks
- Sophie Solar, Emily Blake, Sithembile Chithenga, Mefruz Haque, Anitra Denson, Renee Zell, Jennifer Steppe, Anil Mangla, Preetha Iyengar
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s66
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: At the start of the COVID-19 pandemic, the DC Department of Health (DC Health) mandated new case reporting for early outbreak detection: (1) weekly healthcare personnel (HCP) absenteeism line lists indicating staff absent for confirmed or suspected SARS-CoV-2, (2) daily line lists of all SARS-CoV-2–positive inpatients, and (3) hospital contact tracing. Between March 27, 2020, and December 31, 2020, DC Health detected 36 confirmed and 14 suspected hospital outbreaks, of which only 18% (8 confirmed and 1 suspect) were known to the affected hospital. DC Health learned which outbreaks warranted early or aggressive intervention by tracking outbreak characteristics across its jurisdiction. This allowed prioritization of during surges when it was difficult for DC Health and hospital staff to investigate every outbreak. Methods: Potential outbreaks in short-stay and inpatient rehabilitation hospitals were flagged after identifying SARS-CoV-2 hospital-onset (HO) inpatients or staff clusters on line lists. Variables of interest in line lists included specimen collection and hospital admission dates, units or departments, and patient contact. Facility contact tracing by infection preventionists further verified epidemiological links among cases. Outbreak details were systematically tracked in a locally developed REDCap database and were analyzed if they had an initial case, outbreak start date, or an investigation start date in 2020. Frequency procedures, SQL statements, and date calculations were computed using SAS Enterprise Guide version 8.3 software. Results: Confirmed outbreaks had an average of 6.92 (range, 0–32) HCP and 2.58 (range, 0–22) patient cases, with 69% being confirmed-HO cases and 31% probable HO. Moreover, 53% of confirmed outbreaks occurred in the following departments: cardiac, behavioral health, intensive care, and environmental services (EVS)/facilities. All of these departments had recurrent outbreaks. Behavioral health, medical and cardiac units had the highest number of patient cases. On average, confirmed outbreak investigations lasted 24.6 days, with outbreaks prolonged in the ICU (40.25 days) and the medical unit (37.67 days). Top triggers for investigations ultimately classified as confirmed outbreaks were (1) positive symptomatic HCP, (2) confirmed-HO cases, and (3) exposures from positive HCP. Conclusions: The dynamic nature of COVID-19 created challenges in detecting and responding to hospital outbreaks. Developing a low-resource outbreak tracking system helped identify outbreak types and triggers that warranted early or aggressive interventions. Understanding the characteristics of hospital outbreaks was critical for maximizing infection control resources during surges of infectious disease outbreaks, such as COVID-19. Hospitals or local health departments could adapt this system to meet their needs.
Funding: None
Disclosures: None
Poster Presentation - Oral Presentation
Antibiotic Stewardship
Susceptibility results discrepancy analysis between NHSN Antibiotic Resistance (AR) Option and laboratory instrument data
- Youssoufou Ouedraogo, Christopher Evans, Daniel Muleta, Christopher Wilson
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s66
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: The NHSN Antibiotic Resistance (AR) Option can serve as a useful tool for tracking antibiotic-resistant infections and can aid in the development of inpatient antibiograms. We recently described the frequency of antibiotic suppression in NHSN AR Option data. In this analysis, we describe the effects of suppression on practical uses of the NHSN AR Option, specifically selected agent antibiogram development, and detection of reportable conditions. Methods: Antibiotic susceptibility data were collected from the NHSN AR Option and commercial automated antimicrobial susceptibility testing instruments (cASTI) from 3 hospital networks. Data were obtained from January 1, 2017, to December 31, 2018. The clinical susceptibility data for third-generation cephalosporins and carbapenems against carbapenem-resistant Enterobacterales (CRE), Pseudomonas aeruginosa, and Acinetobacter baumannii were included. Susceptibility results were defined as suppressed when susceptibility results were observed from the laboratory instrument but not from NHSN data. For the overall percentage susceptibility estimation, isolates with <30 susceptibility results were excluded. Percentage susceptibility of NHSN results were compared to their counterparts from cASTI. Results: Of the 852 matched isolates in the primary analysis, 804 had at least 1 suppressed result. Of the 804 isolates, 16.9% were P. aeruginosa, 67.3% by E. coli, and 11.1% by Klebsiella spp. The following pathogen–drug combinations had no difference observed in the percentage susceptible between the 2 systems: ceftazidime tested against P. aeruginosa, ceftriaxone tested against Klebsiella spp, ertapenem tested against Klebsiella spp, imipenem tested against E. coli and P. aeruginosa, and meropenem tested against P. aeruginosa. Significant differences were observed for the following drugs tested against E. coli: ceftazidime (11.1%), cefotaxime (8.6%), and ceftriaxone (8.3%). In the NHSN AR Option, the following isolates showed suppressed results related to their phenotypic case definition: 17 (3%) CRE isolates, 7 (28%) carbapenem-resistant Acinetobacter baumannii (CRAB) isolates, 511 (93.2%) extended spectrum β-lactamase (ESBL) isolates, and 94 (66.7%) carbapenem-resistant Pseudomonas aeruginosa (CRPA) isolates. Conclusions: For select isolates, notably E. coli, we observed a large difference in the percentage of susceptible isolates reported into the NHSN AR Option compared to the cASTI data. This difference significantly limits the ability of the AR Option to create valid antibiograms for select pathogen–drug combinations. Moreover, significant numbers of CRAB, ESBL, and CRPA isolates would not be identified from NHSN AR Option because of suppression. This finding warrants the need for antimicrobial stewardship teams to regularly assess the impact of selective reporting in identifying pathogens of public health importance.
Funding: None
Disclosures: None
Factors associated with antimicrobial drug prescription among inpatient dogs and cats at an academic veterinary hospital
- Emma Price, Claire Fellman, Annie Wayne, Manlik Kwong, Kirthana Beaulac, Shira Doron
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s67
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Widespread antimicrobial use in dogs and cats drives antimicrobial resistance in both animals and humans. Knowledge of the factors associated with antimicrobial use is limited in veterinary medicine. We examined factors associated with antimicrobial drug prescription among inpatient dogs and cats at an academic veterinary hospital. Methods: A veterinary-adapted observational medical outcomes partnership common data model was utilized to extract demographic, clinical, and prescription data from the electronic medical record system in this descriptive observational study. Using generalized estimating equations, we assessed the association between demographic and clinical factors and systemic antimicrobial drug prescription among inpatient dogs and cats at a small-animal teaching hospital between 2018 and 2020. Results: Across 11,685 dogs with 14,328 admissions (mean age, 7.4 years; 47% females), the following factors were associated with increased odds of any antimicrobial drug prescription: female, longer admission, a history of chemotherapy within 30 days of hospital admission, surgery upon admission or within the last 30 days, urinary catheterization, ICU admission, and oxygen support. In 3,371 cats with 4,088 admissions (mean age, 8.6 years; 39% females), the following factors were associated with increased odds of any antimicrobial drug prescription: female, longer admission, increased age (>8 years), admission into the ICU, surgery upon admission, and feline that did not require oxygen support or urinary catheterization. Conclusions: This study identifies multiple patient and clinical factors associated with increased risk of antimicrobial drug use in inpatient dogs and cats that can inform veterinary antimicrobial stewardship efforts and may be useful for antimicrobial use benchmarking on an institutional or multi-institutional scale.
Funding: None
Disclosures: None
Prior cultures predict subsequent susceptibility in patients with recurrent urinary tract infections
- Marissa Valentine-King, Barbara Trautner, Roger Zoorob, George Germanos, Jason Salemi, Kalpana Gupta, Larissa Grigoryan
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s67
-
- Article
-
- You have access Access
- Open access
- Export citation
-
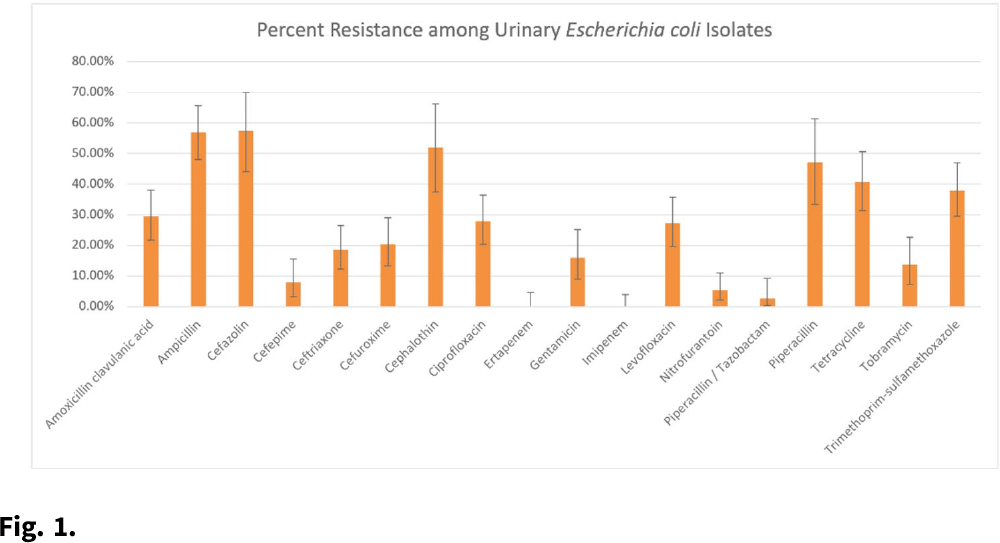
Background: Patients with recurrent urinary tract infections (rUTI) experience frequent exposure to antimicrobial regimens, leaving them at higher risk for developing antibiotic resistance. Little information on the prevalence of antibiotic resistance among patients with rUTI has been published. Although the IDSA recommends using a prior culture to guide empiric treatment, studies have not examined the predictive ability of a prior culture among patients meeting rUTI criteria. We constructed an antibiogram and evaluated test metrics, including sensitivity, specificity, and positive predictive value (PPV) and negative predictive values (NPV) of a prior culture (any organism), on predicting resistance (PPV) or susceptibility (NPV) of a future culture among patients with uncomplicated rUTI in an outpatient setting. Methods: We retrospectively extracted electronic health record data from outpatients aged ≥18 years who had an ICD-10 code for cystitis listed twice in 6 months or thrice in 12 months between November 1, 2016, and December 31, 2018. Patients sought care at either urology or primary care practices within an academic medical center in Houston, Texas. Patients with functional or structural abnormalities of the genitourinary tract, signs or symptoms of pyelonephritis, or pregnancy were excluded. Antibiogram data were reported for uropathogens with ≥30 isolates, and intermediate results were considered resistant. Test metrics and Bayes’ PPV and NPV were calculated using standard formulas. Results: We included 597 visits from 232 unique patients. Most were White (63%) and female (92%), and the cohort had a median age of 58 (IQR, 41–68). Among 310 rUTI episodes with a urine culture, 189 (61%) had at least 1 uropathogen isolated, and Escherichia coli (n = 130, 66%) was most common among all 196 uropathogens. E. coli isolates had >20% resistance to 10 of 18 antibiotics (Fig. 1). E. coli resistance to ciprofloxacin was 27.9%, resistance to nitrofurantoin was 5.5%, and resistance to trimethoprim-sulfamethoxazole was 38.0%. The PPVs for predicting resistance were highest for ceftriaxone (0.86; 95% CI, 0.60–0.96), ciprofloxacin (0.84; 95% CI, 0.63–0.94), and levofloxacin (0.84; 95% CI, 0.63–0.94). NPVs of resistance were highest for gentamicin (0.97; 95% CI, 0.83–1.00), ceftriaxone (0.94; 95% CI, 0.86–0.98), and cefepime (0.94; 95% CI, 0.84–0.98), whereas NPVs for cefuroxime, ciprofloxacin, levofloxacin, and nitrofurantoin were all >0.83. Conclusions: We detected considerable antibiotic resistance among patients with rUTI to commonly prescribed antibiotics. Prior urine culture susceptibility demonstrated moderate-to-high PPVs for predicting future resistance to ceftriaxone and fluoroquinolones as well as high NPVs for several cephalosporins and fluoroquinolones, which could inform empiric prescribing choices.
Funding: This investigator-initiated research study was funded by Rebiotix, a Ferring Company.
Disclosures: None
The effect of gender bias on acceptance of antibiotic stewardship recommendations by clinical pharmacists
- Valerie Vaughn, Daniel Giesler, Adamo Brancaccio, Daraoun Mashrah, Katie Sandison, Chaorong Wu, Jennifer Horowitz, Linda Bashaw, Adam Hersh
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s67-s68
-
- Article
-
- You have access Access
- Open access
- Export citation
-
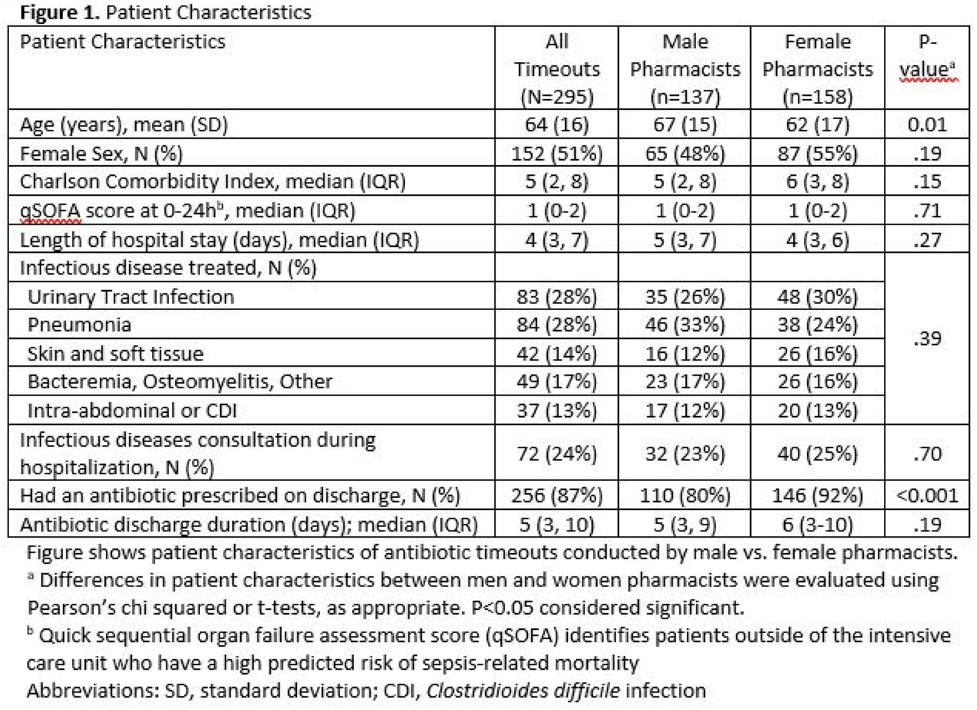
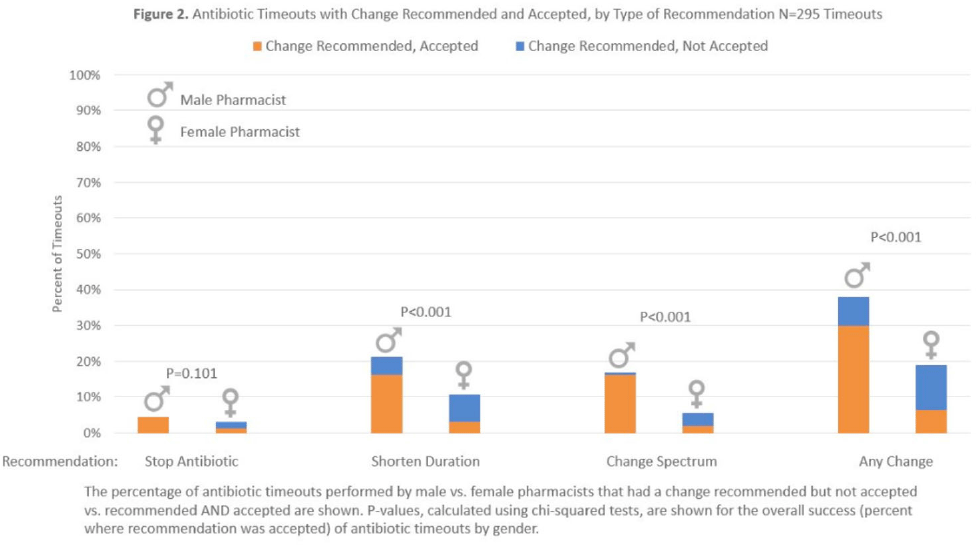
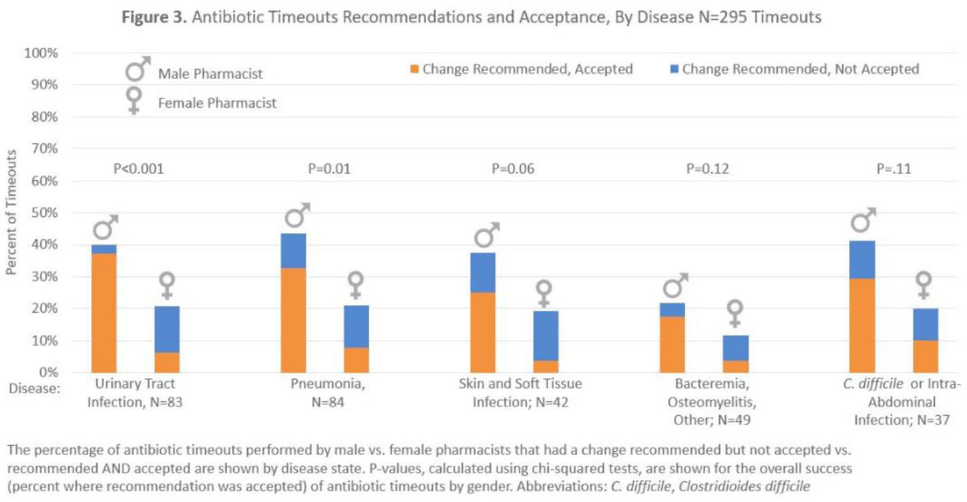
Background: Clinical pharmacists are a critical part of antibiotic stewardship. Stewardship often relies on relationships and persuasion, which may be affected by gender bias. Thus, we aimed to assess the association of sex with the acceptance of antibiotic stewardship recommendations. Methods: Between May and October 2019, medicine pharmacists at single hospital reviewed patients on antibiotics and–when a discharge was anticipated–led an antibiotic discussion (or “timeout”) prior to discharge. To explore differences in antibiotic timeout effectiveness by gender, we assessed the association of pharmacist sex with suggestion and acceptance of antibiotic changes using logistic regression controlling for patient characteristics. We also assessed whether hospitalist sex was associated with or moderated the effect of pharmacist sex on acceptance of timeout recommendations. Results: Between May 1, 2019, and October 31, 2019, pharmacists conducted 295 timeouts (patient characteristics in Fig. 1). Overall, 54% of timeouts were conducted by 12 female pharmacists and the remaining 46% were conducted by 8 male pharmacists. Overall, 82 (29%) of 295 timeouts resulted in a pharmacist recommending an antibiotic change, and male pharmacists were more likely to recommend a change: 52 (38%) of 137 versus 30 (19%) 158 (P Conclusions: In this discharge antibiotic intervention, timeouts conducted by women were less likely to result in an antibiotic change than those conducted by men. The difference in effectiveness resulted both from female pharmacists being less likely to recommend a change and from hospitalists being less likely to accept recommendations from a female pharmacist. These findings suggest that gender bias may play a role acceptance of antibiotic stewardship recommendations, which could affect antibiotic use, pharmacist job satisfaction, and patient outcomes.
Funding: None
Disclosures: None
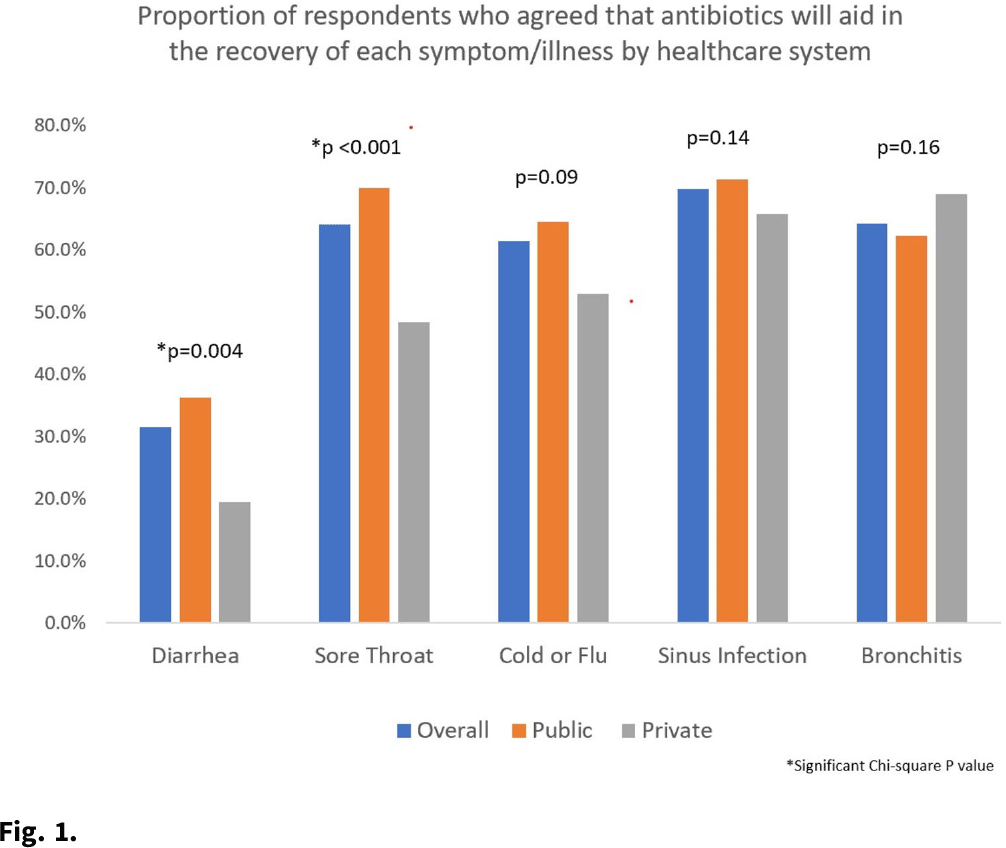
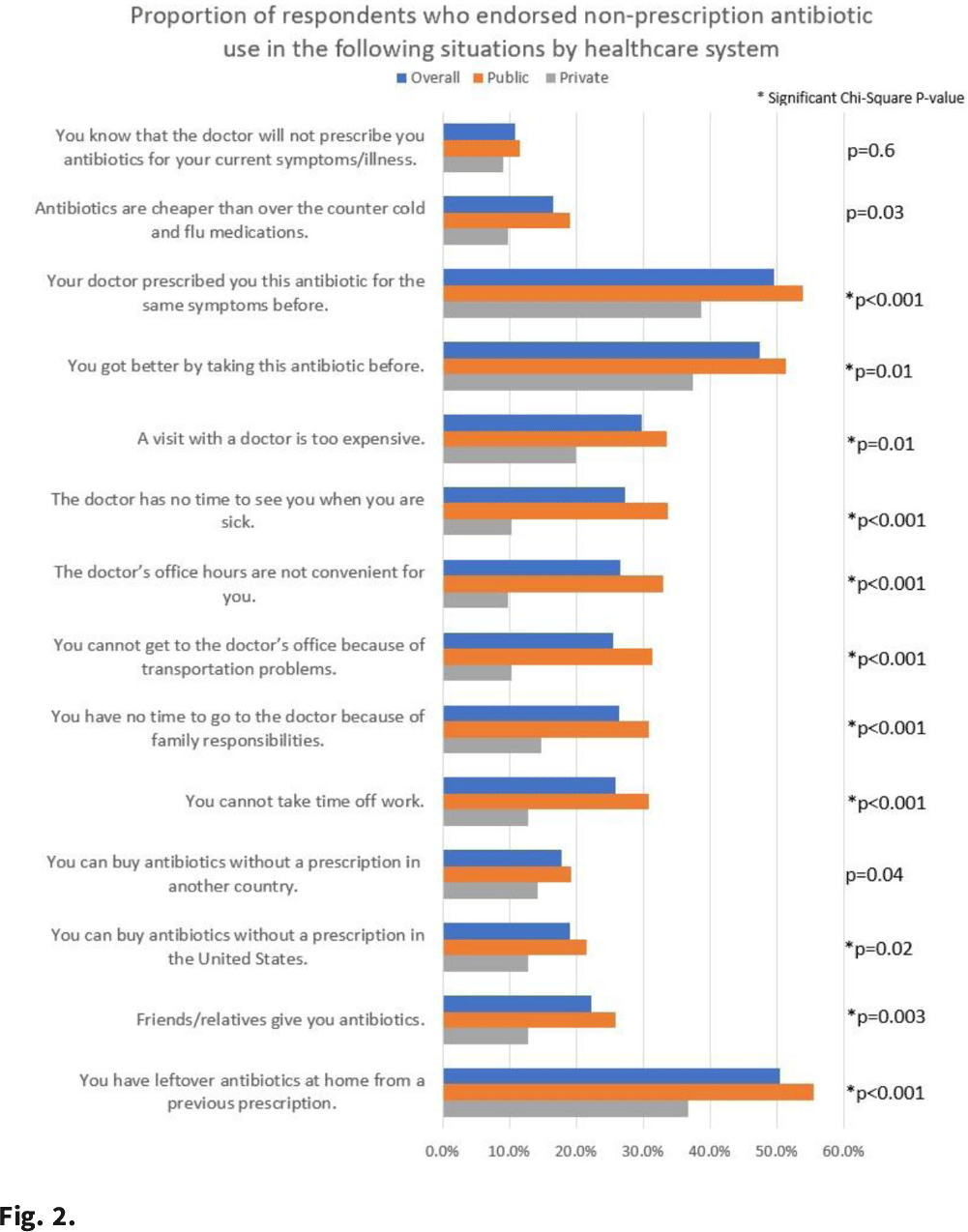
Identifying symptoms/illnesses and situations that predispose outpatients to use antibiotics in two healthcare systems
- Lindsey Laytner, Larissa Grigoryan, Barbara Trautner, Osvaldo Alquicira, Juanita Salinas, Michael Hansen, Roger Zoorob, Fareed M. Khan
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s68-s69
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Taking antibiotics outside the guidance of a clinician (nonprescription use) is a potential safety issue and runs counter to antibiotic stewardship efforts. We identified the symptoms and illnesses and situations that may predispose patients to take antibiotics, and we compared these findings between patients attending public primary care clinics and private emergency departments. Methods: A cross-sectional survey was conducted between January 2020 and March 2021 in 6 primary care clinics and 2 emergency departments in the United States. We queried patients about 5 symptoms and illnesses (Fig. 1) and 14 situations (Fig. 2) to investigate whether these would lead the patients to take antibiotics without a prescription. We used the χ2 test to compare the symptoms and illnesses and situations between the respondents from public and private healthcare systems. We set the P value for significance at <.025. Results: In total, the survey had 564 respondents (median age, 49.7 years; range, 19–92), and 72% were female. Most respondents identified as either Hispanic or Latina/Latino (46.6%) or African American or Black (33%), followed by White (15.8%), and other (4.6%). Most respondents had visited public clinics (72%). The most common insurance status for our respondents included Medicaid or county financial assistance program (56.6%), followed by private insurance or Medicare (36.7%) and self-pay (6.7%). In public primary care clinics, only 23% had private insurance or Medicare compared to 72.9% in private emergency departments. Of those surveyed, 69% agreed that antibiotics would improve the recovery from sinus infections, followed by bronchitis (64%), sore throat (64%), cold/flu (61.4%), and diarrhea (31.5%). The proportions of respondents who believed that antibiotics would improve the recovery from diarrhea (36.2% vs 19.4%; P = .004) and sore throat (59.9% vs 48.4%; P < .001) were significantly higher among public versus private outpatient respondents. We did not find significant differences for cold/flu, sinus infection, or bronchitis between these 2 healthcare systems (Fig. 1). In 11 of the 14 situations, patients in public clinics were more likely to report a likelihood of using nonprescription antibiotics than the patients visiting the private emergency rooms (Fig. 2). Conclusions: Future stewardship interventions should be aware of the symptoms and illnesses and situations that may influence outpatients to take nonprescription antibiotics. Addressing modifiable factors (eg, leftover antibiotics, antibiotics given by friends or family, and antibiotics available without a prescription in stores or markets) may also curtail these unsafe practices and reduce antibiotic resistance.
Funding: None
Disclosures: None
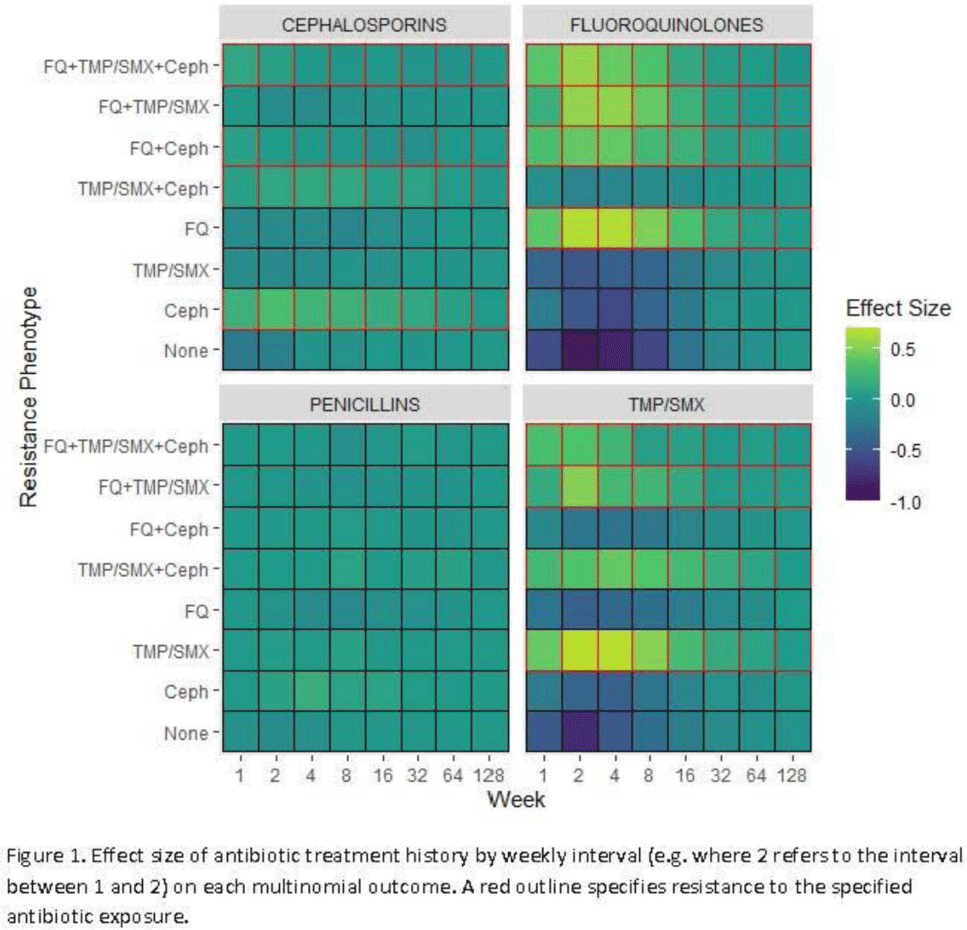
Using machine learning to predict antibiotic resistance to support optimal empiric treatment of urinary tract infections
- Ben Brintz, McKenna Nevers, Matthew Goetz, Kelly Echevarria, Karl Madaras-Kelly, Matthew Samore
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s69
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Antibiotic resistance is pervasive in the Veterans’ Affairs (VA) healthcare system, with rates of fluoroquinolone and trimethoprim–sulfamethoxazole (TMP/SMX) resistance approaching 30% in E. coli urinary isolates. The efficacy of antimicrobial treatment is critically dependent on the susceptibility of the infecting pathogen; however, prescription decisions are often made empirically in practice. We analyzed susceptibility profiles of enteric gram-negative rods (Enterobacterales) from clinical urine cultures collected from ambulatory patients receiving care in VA clinics and emergency departments. Our goals were (1) to develop a predictive model to support choice of empiric antibiotics pending results of susceptibility testing and (2) to examine the relationship between past antibiotic exposures and susceptibility profiles to enhance understanding of antibiotic selective pressure. Methods: We obtained 265,076 positive cultures from 157,422 unique patients from 2015 to 2020. We trained random forest multinomial classifiers to estimate the risk of a positive urine culture isolate being resistant to the multinomial outcome: fluoroquinolone, TMP–SMX, cephalosporin, or any combination of these 3 agents. Data sources evaluated for model generation included demographics, comorbidities, trend and seasonal terms, treatment history for multiple antimicrobial treatments summarized using number of prescriptions in weekly intervals, and sample history summarized by number of resistant and susceptible cultures in weekly intervals. Using 5-fold cross validation, we assess the performance of the clinical prediction using the area under the receiver operating characteristic curve (AUC) for each multinomial outcome. In addition to prediction, we modeled the direct effect of treatment on resistance using multinomial group lasso (MGL). This method allows variable selection in variable groupings, such as all variables related to the fluoroquinolone treatment history, which allowed us to assess the effect of a patient’s complete course of treatment on resistance. Results: In cross-validation analysis, our random forest model was best at predicting outcomes with fluoroquinolone resistant phenotypes compared to non–fluoroquinolone-resistant phenotypes (Table 1). From MGL, we found that having a prescription for fluoroquinolone treatment 4–8 weeks prior to a urinalysis was positively associated with fluoroquinolone resistance and negatively associated with fluoroquinolone susceptible phenotypes (Fig. 1). Conclusions: Our results show that a patient’s sample and treatment history are highly predictive of a future resistance. Fluoroquinolone treatment is especially associated with increased risk of fluoroquinolone single- and multidrug resistances. A history of either fluoroquinolone or trimethoprim-sulfamethoxazole (TMP-SMX) treatment is a stronger indicator of a future resistant phenotype than cephalosporin or penicillin.
Funding: None
Disclosures: None

Private practice dentists improve antibiotic use after dental antibiotic stewardship from infectious diseases experts
- Debra Goff, Julie Mangino, Elizabeth Trolli, Douglas Goff
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s70
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Dentists prescribe ~25.7 million antibiotic prescriptions annually. Private practice dentists (PPDs) represent 80% of US dentists who need to implement dental antimicrobial stewardship. We conducted a prospective cohort study of PPDs comparing appropriateness of antibiotic use before and after dental AS education. Methods: PPDs were invited to participate in this study. In phase 1 (pre-education), we collected 3 months (June–August 2019) of retrospective antibiotic use data (indication, dose, duration, penicillin allergy history) and number of dental procedures. We also conducted a preliminary survey to assess baseline antimicrobial stewardship knowledge. In phase 2 (education), PPDs attended 4 televideo education sessions (March–May 2021) taught by an infectious disease– antimicrobial stewardship (ID-AS) pharmacist and physician. In phase 3 (posteducation), we prospectively collected 3 months (June–August 2021) of antibiotic use data (as in phase 1), using an online database with ongoing feedback. In phase 4, we conducted antibiotic use audit and feedback to PPDs after the survey, and we solicited recommendations to reach more PPDs. The Student t test was used for statistical analyses. Results: Study participants comprised 15 PPDs: 2 oral maxillofacial surgeons, 6 periodontists, 4 endodontists, and 3 general dentists. Among them, 10 had been in practice >20 years. The presurvey revealed that 14 were unfamiliar with dental antimicrobial stewardship. All prescribed clindamycin (25% for nonpenicillin allergy), and standard antibiotic duration ranged from 5 to 14 days based on dental school training. In phase 3, despite more procedures, overall antibiotic use and duration decreased, and the use of clindamycin, quinolones, and prophylaxis for joint implant patients, also decreased. Appropriate use improved from 22% to 95%. Postsurvey responses on perceived value of antimicrobial stewardship education were 100% positive, with recommendations to make antimicrobial stewardship a required annual continuing education, similar to opioid continuing education. Study participants invited the ID-AS experts to teach an additional 150 PPDs to date via established PPD study clubs to expand dental antimicrobial stewardship across the United States. Conclusions: After learning dental antimicrobial stewardship guidance from ID-AS experts, PPDs rapidly optimized antibiotic prescribing behavior. PPDs identified their established study clubs as a forum to quickly expand dental antimicrobial stewardship training by ID-AS experts throughout the United States.
Funding: MERCK
Disclosures: None

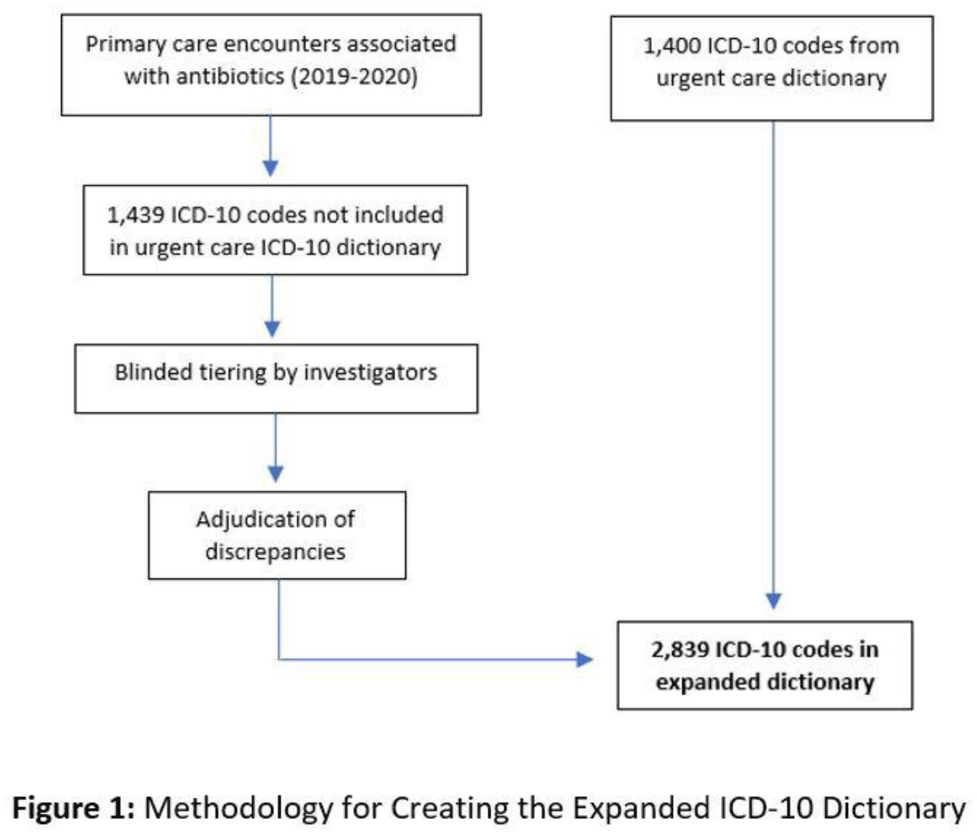
Metrics in outpatient stewardship: Is more always better?
- Natalia Medvedeva, David Ha, Sharon Onguti, Emily Rosen, Emily Mui, Sean Pearce, Alex Schneider, Amy Chang, Adam Hersh, Eddie Stenehjem, Marisa Holubar
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s70-s71
-
- Article
-
- You have access Access
- Open access
- Export citation
-
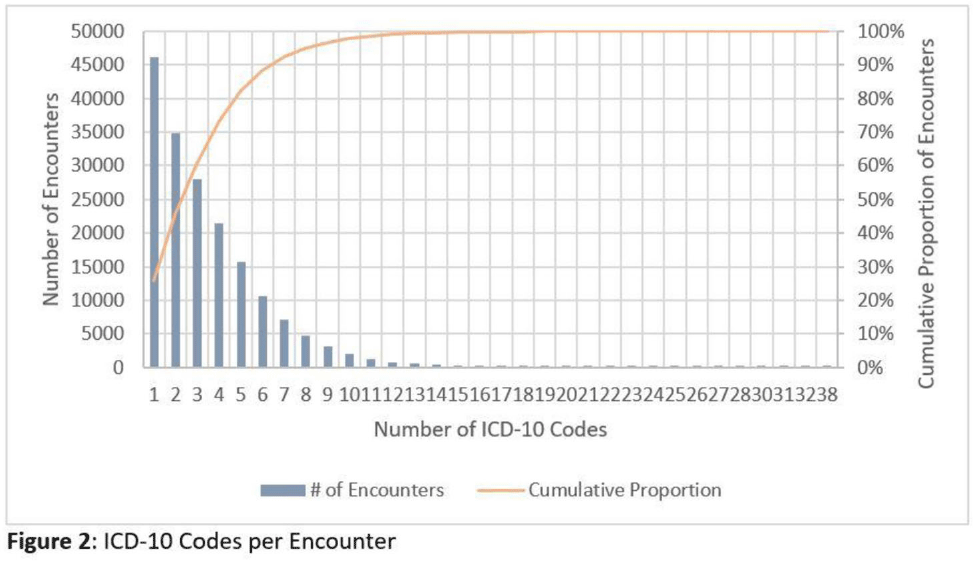
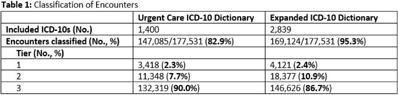
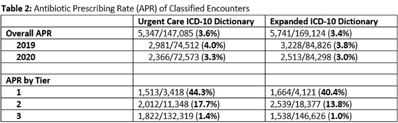
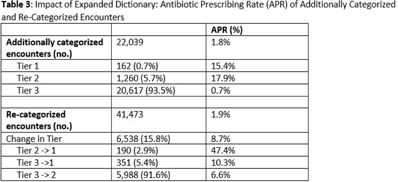
Background: Emerging evidence supports the use of billing data to identify stewardship targets in primary care. Standardizing an approach to antibiotic prescribing rate (APR) calculations could facilitate external benchmarking. Methods: Using methodology and an ICD-10 dictionary validated in urgent care clinics,1 we created an expanded ICD-10 dictionary to incorporate additional ICD-10 codes from primary care associated with antibiotic prescriptions (Fig. 1). We then compared antibiotic prescribing rates using the urgent care and expanded dictionaries. We included all primary care visits from 2019 to 2020 and extracted ICD-10 codes and antibiotic order data. Using the urgent care and expanded ICD-10 dictionary, we classified each encounter by prescribing tier based on whether antibiotics are almost always (tier 1), sometimes (tier 2), or almost never (tier 3) indicated. For encounters with ICD-10s in multiple tiers, we chose the lowest tier. For multiple ICD-10 codes within the same tier, we chose the first extracted ICD-10 code. We calculated antibiotic prescribing rates as the proportion of encounters associated with ≥ 1 antibacterial prescription. This quality improvement project was deemed non–human subjects research by the Stanford Panel on Human Subjects in Medical Research. Results: The urgent care dictionary has 1,400 ICD-10 codes. We added 1,439 ICD-10 codes derived from primary care encounters to create the expanded ICD-10 dictionary (8.5% tier 1, 9.1% tier 2, and 82.4% tier 3) (Fig. 1). We identified 177,531 encounters; 74% had ≥ 2 associated ICD-10 codes (Fig. 2). In total, 147,085 encounters (82.9%) were classified into a tier using the urgent care dictionary. An additional 22,039 encounters were classified with the expanded dictionary (Table 1). Most added encounters were tier 3 with low 0.7% APR (Tables 1 and 3). In total, 41,473 (28.2%) encounters were classified differently depending on the ICD-10 dictionary used, most commonly changing from tier 3 to tier 2 without an increase in overall tier 2 antibiotic prescribing rate (Tables 2 and 3). Overall antibiotic prescribing rates were similar when using either the urgent care or expanded ICD-10 dictionary (Table 2). Conclusions: The expanded ICD-10 dictionary allowed for classification of more encounters in primary care; however, it did not meaningfully change antibiotic prescribing rates. Antibiotic prescribing rates were likely diluted by classifying more encounters without identifying an associated increase in antibiotic prescribing. A more sophisticated classification system may help to accommodate the diversity and volume of ICD-10 codes used in primary care.
1. Stenehjem E, et al. Clin Infect Dis 2020;70:1781–1787.
Funding: None
Disclosures: None
Evaluation of periprocedure antibiotics and infection-related hospitalizations after transrectal prostate biopsies
- Tenley Ryan, Neena Thomas-Gosain, Jane Eason, Hanna Akalu, Navila Sharif, Jessica Bennett
-
- Published online by Cambridge University Press:
- 16 May 2022, p. s71
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background: Prostate cancer is the leading cancer diagnosis and the second leading cause of cancer deaths in men. Definitive diagnosis is made by prostate biopsy. This procedure poses a risk of infection and, rarely, sepsis. Studies have found the incidence of symptomatic urinary tract infection (UTI) after biopsy to be 2%–3%, and the rate of infection-related hospitalization (IRH) to be 0.6%–4.1%. An initial review at our facility found the IRH rate to be 3.7%. The primary purpose of this study was to determine the incidence of IRH following prostate biopsy in patients at the Memphis VA Medical Center (VAMC) after initial review and education. Methods: All transrectal prostate biopsies performed at the Memphis VAMC from October 2017 through May 2021 were analyzed. Patients were excluded if they had a spinal cord injury or concomitant procedure. The primary outcome was IRH occurring within 30 days of the procedure. Variables collected included risk factors, antibiotic choice and duration, and details of postprocedural infections. Analyses were performed on a per-procedure basis. Results: Overall, 601 procedures were identified; 13 were excluded, for a total of 588 transrectal prostate biopsies on 533 patients. All patients were given antibiotics. Oral antibiotics alone were provided for 306 procedures (52%) for an average duration of 3 days. A combination of both oral and intramuscular antibiotics were provided for 282 (48%) procedures. The most common oral antibiotics used were cefuroxime (538, 91.4%), ciprofloxacin (17, 2.9%), amoxicillin–clavulanate (16, 2.7%), and sulfamethoxazole–trimethoprim (12, 2%). Intramuscular antibiotics included ceftriaxone (263, 93.3%) and gentamicin (19, 6.7%). An infectious complication occurred in 29 patients (4.9%): 26 (3.4%) were urogenital and 5 (0.8%) required hospitalization. Of the procedures complicated by a postprocedure infection, 22 (75.9%) received an oral antibiotic alone, 21 (95.4%) of which were cefuroxime, and 7 (24.1%) received both an intramuscular and an oral agent. Conclusions: In our initial review, the most common antibiotics used were fluroquinolones, with an average duration of 3 days periprocedure and an IRH rate of 3.7%. These findings were used to reinforce practices compliant with American Urological Association (AUA) guidelines. This follow-up review reveals that the first-line choice changed from fluroquinolones to cephalosporins, with average duration remaining at 3 days. Although the overall infection rate was 4.9%, the IRH rate decreased from 3.7% to 0.8%.
Funding: None
Disclosures: None
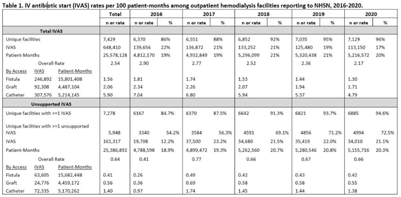
Rates of intravenous antibiotic starts among outpatient hemodialysis patients using NHSN dialysis event reporting, 2016–2020
- William Wilson, Sarah Kabbani, Shannon Novosad, Lucy Fike, Katryna Gouin, Jeneita Bell, Suparna Bagchi, Jonathan Edwards, Ibironke Apata, Susan Cali
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s71-s72
-
- Article
-
- You have access Access
- Open access
- Export citation
-
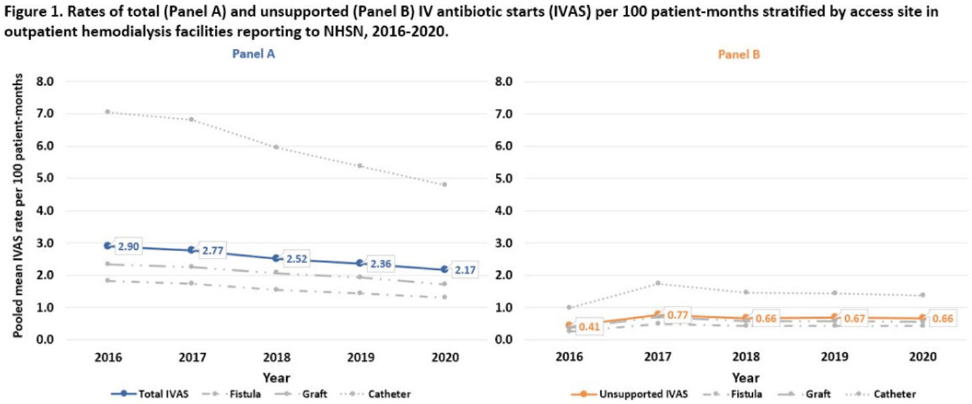
Background: Nearly one-third of patients on hemodialysis receive intravenous (IV) antibiotics annually, but national data characterizing antibiotic use in this population are limited. Using NHSN surveillance data for outpatient dialysis facilities, we estimated temporal changes in the rate of IV antibiotic starts (IVAS) among hemodialysis patients as well as the proportion of IVAS that were not supported by a reported clinical indication. Methods: IVAS events were obtained from the NHSN Dialysis Event module between 2016 and 2020, excluding patients who were out of network, receiving peritoneal or home dialysis, or with unspecified vascular access. IVAS unsupported by documentation were defined as new IVAS without a collected or positive blood culture, pus, redness or swelling event, or an associated clinical symptom. Pooled mean rates of total and unsupported IVAS were estimated per 100 patient months yearly and stratified by vascular access type. Differences in IVAS rates by year were estimated with negative binomial regression. Results: Between 2016 and 2020, 7,278 facilities reported 648,410 IVAS events; 161,317 (25%) were unsupported by documentation (Table 1). In 2016, 3,340 (54%) facilities with ≥1 IVAS event reported an IVAS unsupported by documentation, which increased to 4,994 (73%) in 2020. Total IVAS rates decreased by an average of 8.2% annually (95% CI, 7.1%–9.3%; P < .001). The average annual percentage decrease did not differ significantly by vascular access site. The total IVAS rate was lowest in 2020 (2.17 per 100 patient months; 95% CI, 2.18–2.17). IVAS rates in 2020 were greatest for patients with catheter access (4.79 per 100 patient months; 95% CI, 4.75–4.83), followed by graft (1.71 per 100 patient months; 95% CI, 1.68–1.73), and lowest for patients with fistulas (1.30 per 100 patient months; 95% CI, 1.29–1.31). The overall pooled mean rate of unsupported IVAS was 0.64 per 100 patient months (95% CI, 0.63–0.64), which did not significantly change by year (Fig. 1). Conclusions: Total IVAS rates among outpatient hemodialysis patients have decreased since 2016, and rates among catheter patients remain highest compared to patients with fistulas or grafts. However, unsupported IVAS rates did not change, and the proportion of facilities reporting an unsupported IVAS increased annually. Targeted efforts to engage facilities with unsupported IVAS may help improve accurate reporting and prescribing practices.
Funding: None
Disclosures: None
C. difficile
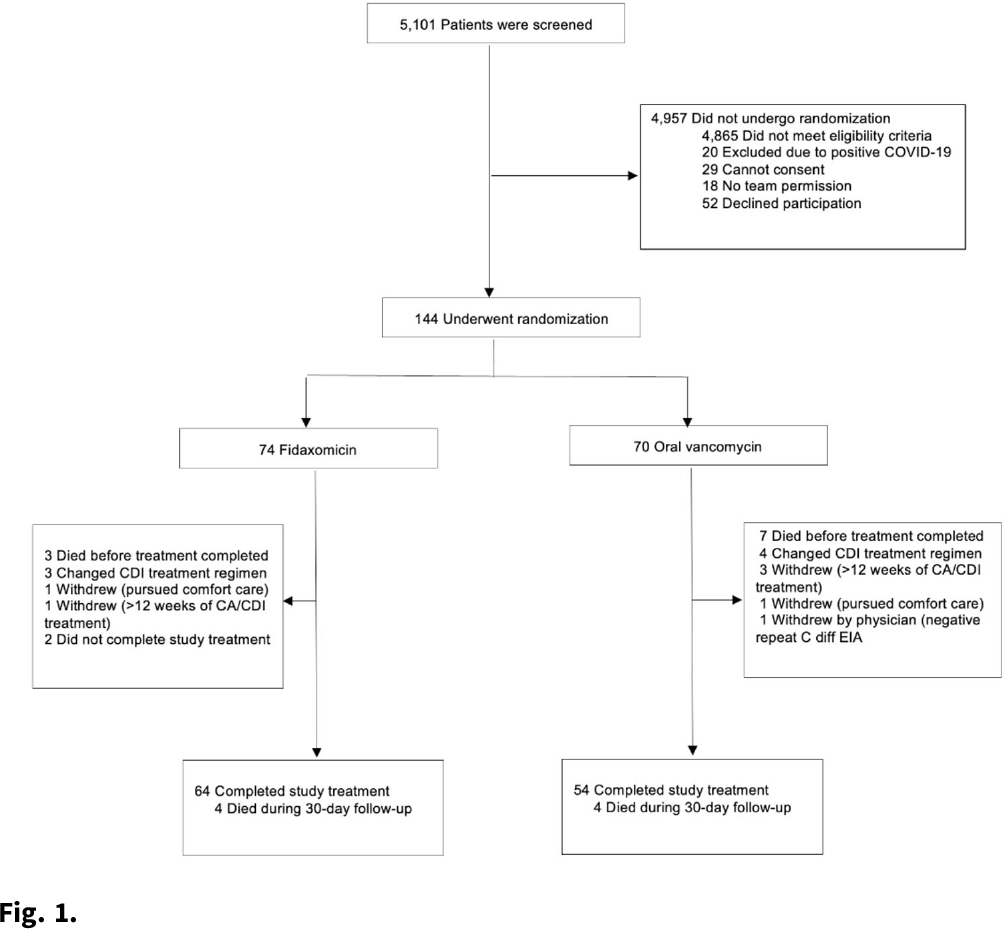
Comparison of fidaxomicin to oral vancomycin for the treatment of Clostridioides difficile infection in hospitalized patients
- A. Krishna Rao, Qianzi Zhao, Jay Krishnan, Justin Bell, Oryan Henig, Jolene Daniel, Kara Sawaya, Owen Albin, John Mills, Lindsay Petty, Kevin Gregg, Daniel Kaul, Anurag Malani, Jason Pogue, Keith Kaye
-
- Published online by Cambridge University Press:
- 16 May 2022, pp. s72-s73
-
- Article
-
- You have access Access
- Open access
- Export citation
-
Background:Clostridioides difficile infection (CDI) is a major source of morbidity and mortality. Even after recovery, recurrent CDI (rCDI) occurs frequently, and concomitant antibiotic use for treatment of a concurrent non–C. difficile infection is a major risk factor. Treatment with fidaxomicin versus vancomycin is associated with similar rate of cure and lower recurrence risk. However, the comparative efficacy of these 2 agents remains unclear in those receiving concomitant antibiotics. Methods: We conducted a randomized, controlled, open-label trial at the University of Michigan and St. Joseph Mercy hospitals in Ann Arbor, Michigan. Patients provided written informed consent at enrollment. We included all hospitalized patients aged ≥18 years with a positive test for toxigenic C. difficile, >3 unformed stools per 24 hours, and ≥1 qualifying concomitant antibiotic with a planned treatment of an infection for ≥5 days after enrollment. We excluded patients with complicated CDI, allergy to vancomycin–fidaxomicin, planned adjunctive CDI treatments, CDI treatment for >24 hours prior to enrollment, concomitant laxative use, current or planned colostomy or ileostomy, and/or planned long-term (>12 weeks) concomitant antibiotic use. Clinical cure was defined as resolution of diarrhea for 2 consecutive days maintained until the end of therapy and for 2 days afterward. rCDI was defined as recurrent diarrhea with positive testing within 30 days of initial treatment. Patients were randomized (stratified by ICU status) to fidaxomicin 200 mg twice daily or vancomycin 125 mg orally 4 times daily for 10 days. If concomitant antibiotic treatment continued >10 days, the study drug continued until the concomitant antibiotic ended. Bivariable statistics included t tests and χ2 tests. Results: After screening 5,101 patients for eligibility (May 2017–May 2021), 144 were included and randomized (Fig. 1). Study characteristics and outcomes are noted in Table 1. Baseline characteristics were similar between groups. Most patients were aged <65 years, were on a proton-pump inhibitor (PPI), and were not in the ICU. The mean duration of concomitant antibiotic was 18.4 days. In the intention-to-treat population, clinical cure (73% vs 62.9%; P =.195), and rCDI (3.3% vs 4.0%; P >.99) were similar for fidaxomicin and vancomycin, respectively. Conclusions: In this study of patients with CDI receiving a concomitant antibiotic, a numerically higher proportion were cured with fidaxomicin versus vancomycin, but this result did not reach statistical significance. Overall recurrence was lower than anticipated in both arms compared to previous studies in which duration of CDI treatment was not extended during concomitant antibiotic treatment. Future studies are needed to ascertain whether clinical cure is higher with fidaxomicin than vancomycin during concomitant antibiotic exposure, and whether extending the duration of CDI treatment reduces recurrence.
Funding: Merck & Co.
Disclosures: None